Can AbbVie’s latest dermatology data at AAD 2026 really secure its post-Humira growth story in immunology?

How do AbbVie Dermatology Data influence the stock?
AbbVie is showcasing 24 abstracts at AAD 2026, underscoring its ambition to dominate immune-mediated skin diseases. The AbbVie Dermatology Data package highlights durable efficacy, long-term safety and quality-of-life gains for Skyrizi (risankizumab) and Rinvoq (upadacitinib) across psoriasis, psoriatic arthritis and atopic dermatitis, plus investigational programs in vitiligo and alopecia areata. On Wall Street, these assets are already seen as the core successors to Humira, with several equity analysts expecting double-digit immunology growth through 2026.
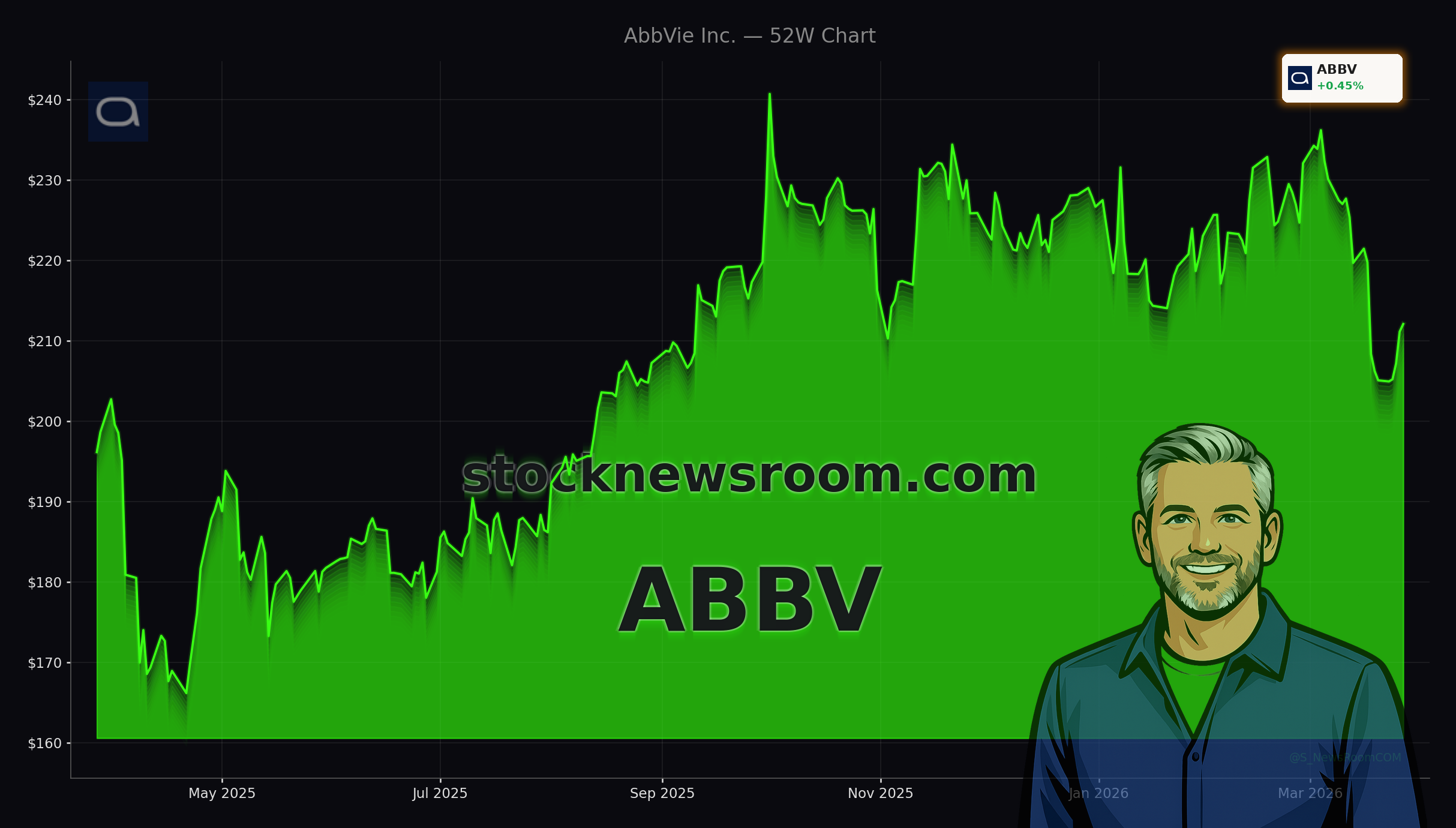
Recent trading confirms that narrative: ABBV outperformed broader indices in Thursday’s session, even as the S&P 500 and Dow slipped. Dividend-focused commentary in the financial press points to AbbVie’s rising payout and robust pipeline as key reasons U.S. income investors keep accumulating shares despite macro volatility.
What stands out in Skyrizi results for investors?
Among the most investor-relevant AbbVie Dermatology Data, five-year KEEPsAKE-1 Phase 3 results showed 88% of psoriatic arthritis patients on Skyrizi had no radiographic progression, reinforcing its role as a long-term structural disease modifier. In plaque psoriasis, a subgroup from the IMMpactful trial in biologic-naïve patients demonstrated higher skin clearance versus deucravacitinib, with PASI 90 rates of 57.3% for Skyrizi versus 22.9% for the comparator at week 16.
For high-burden genital and scalp psoriasis, Phase 4 UnlIMMited data showed 72%–88.9% of genital and 83.3%–100% of scalp patients achieving DLQI 0/1 at week 16. These outcomes have already translated into an FDA label update for Skyrizi, which should support broader dermatology adoption in the U.S. and strengthen AbbVie’s competitive positioning against biologics from peers like Apple-backed digital health partners and large-cap pharma rivals.
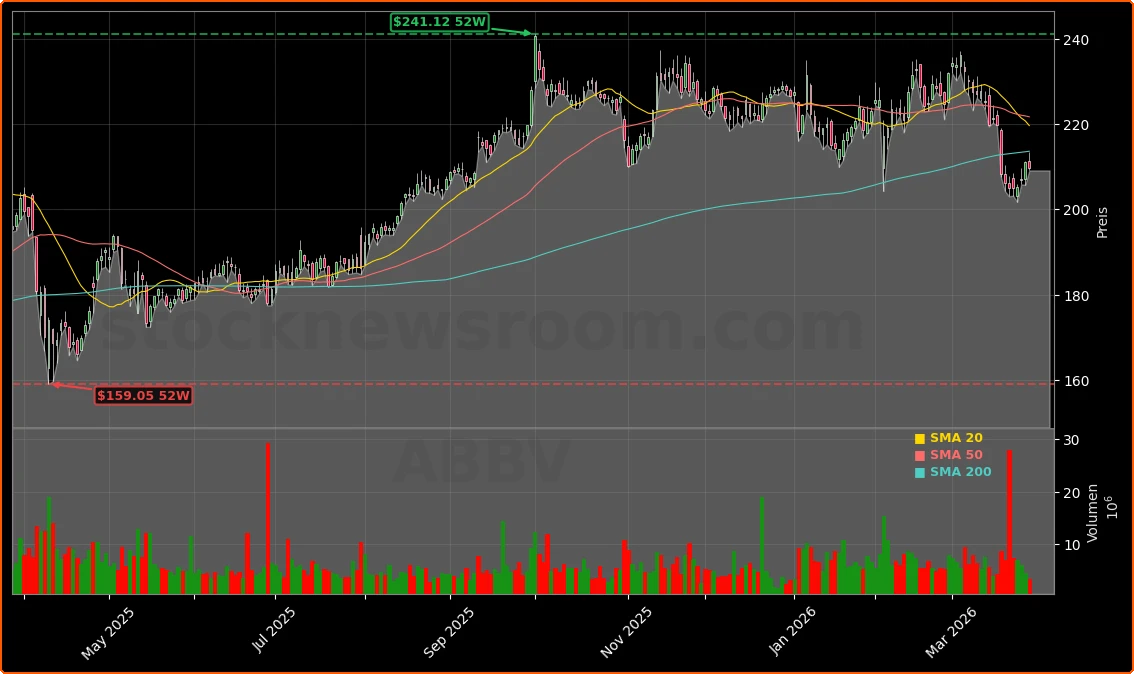
How does Rinvoq expand AbbVie’s skin franchise?
The AbbVie Dermatology Data set also deepens confidence in Rinvoq’s atopic dermatitis profile. Integrated Phase 3 safety analyses across up to six years and 2,683 patients (over 9,000 patient-years) showed consistent adverse event rates across age groups, with no major cardiovascular events in younger cohorts. Real-world AD-VISE data linked minimal disease activity on Rinvoq to superior quality-of-life scores, with 72.3% of those meeting that target reaching DLQI 0/1.
Late-breaking Phase 3 Viti-Up results represent the first systemic Phase 3 data in vitiligo, while UP-AA program data in severe alopecia areata showed more patients on Rinvoq 15 mg or 30 mg achieving meaningful hair regrowth versus placebo. Though not yet FDA approved for these indications, success here would extend AbbVie’s reach into under-served autoimmune dermatology niches and counter momentum at names like NVIDIA and Tesla that currently dominate growth headlines in the S&P 500.
Our robust evidence across multiple clinical trials and real-world data sources for risankizumab and upadacitinib reaffirm AbbVie’s dedication to supporting improved patient outcomes.— Andrew Anisfeld, Ph.D., Vice President, Global Medical Affairs, Immunology, AbbVie
In sum, the AbbVie Dermatology Data presented at AAD 2026 reinforce Skyrizi and Rinvoq as central pillars of the post-Humira story, support expectations for continued dividend increases, and give U.S. investors clear milestones to watch as additional 52-week and Phase 3 readouts emerge over the rest of 2026.