Can the CRISPR Therapeutics Pipeline in cardiology turn today’s modest gene-editing revenues into a multi-billion-dollar growth story?

Is Wall Street underpricing CRISPR Therapeutics?
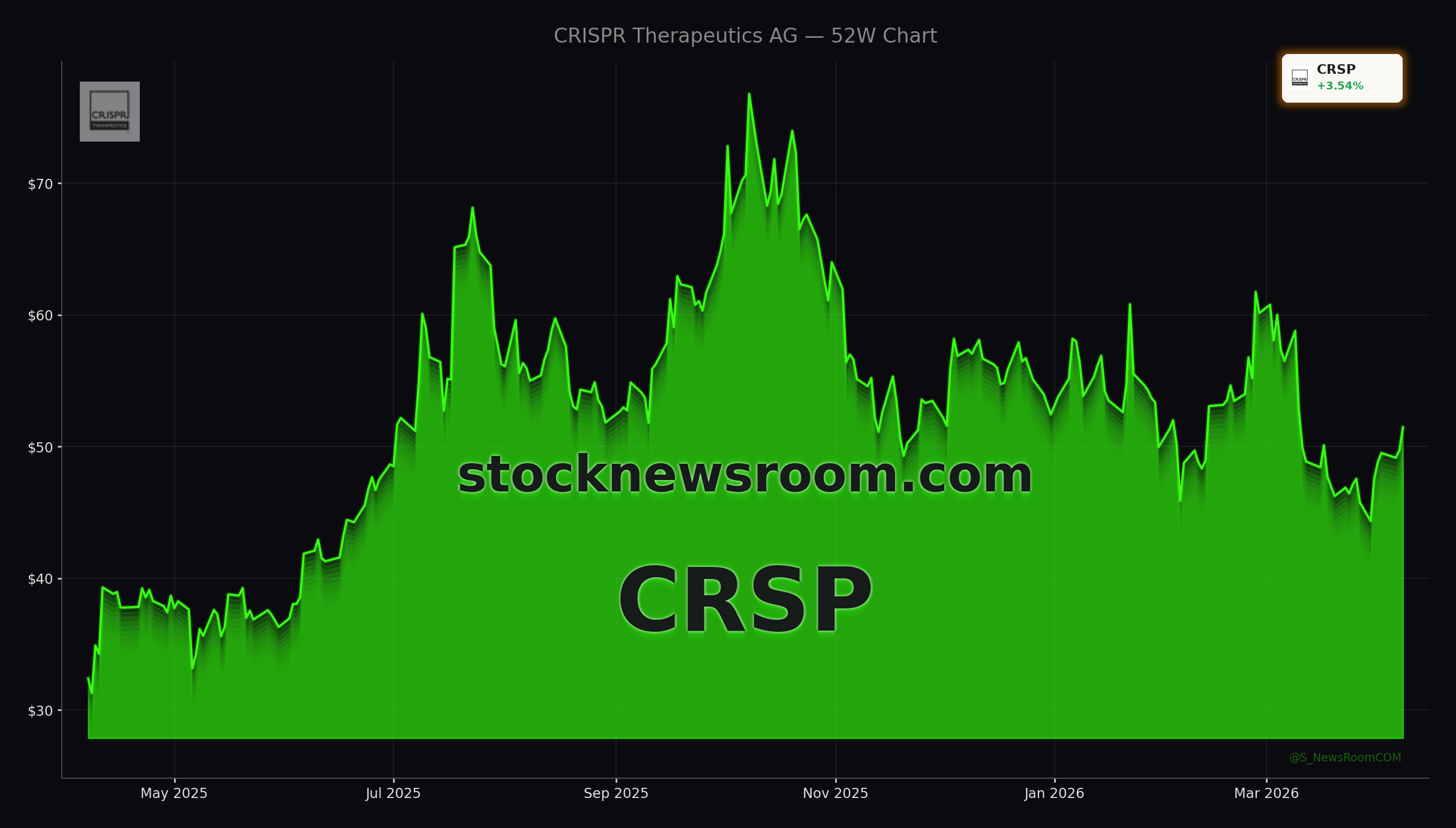
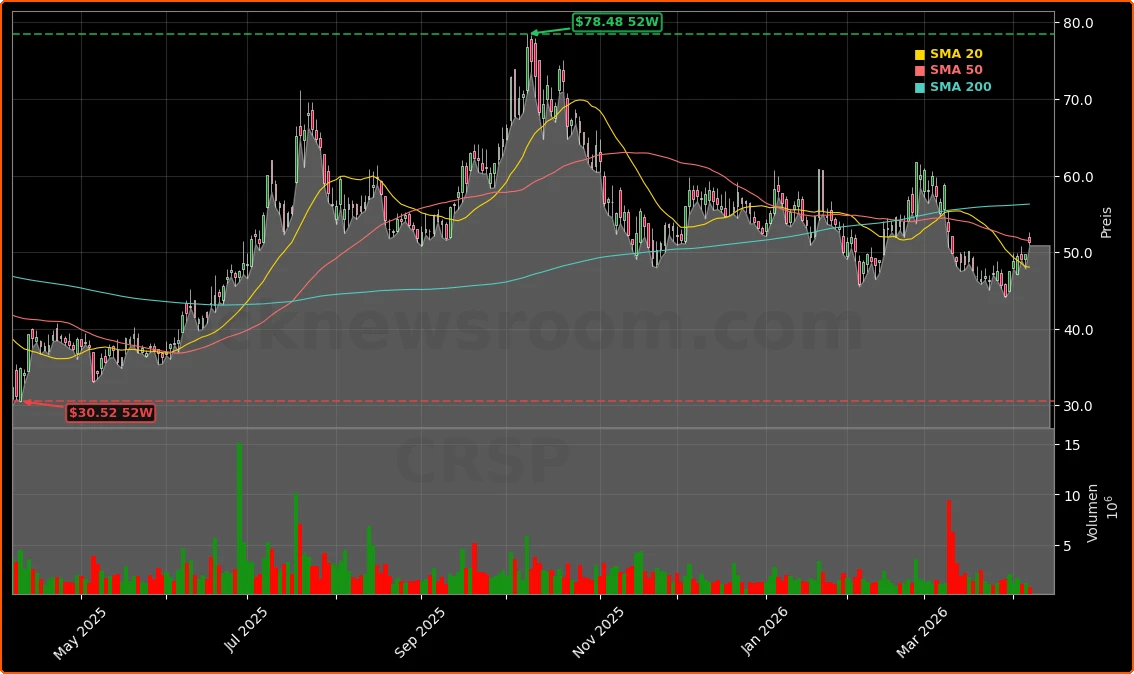
CRISPR Therapeutics has badly trailed the S&P 500 over the last five years, as rising rates and risk-off sentiment punished unprofitable biotech names. Despite launching its first approved gene-editing therapy CASGEVY for blood disorders in partnership with Vertex, commercial revenue remains modest and far from justifying a multi-billion-dollar market cap on its own. The bull case increasingly hinges on the breadth and quality of the CRISPR Therapeutics Pipeline, and cardiology is emerging as one of the most important value drivers.
At a roughly mid-single-digit billion-dollar valuation and a share price near $51, the stock sits well below optimistic analyst targets. Mizuho Securities recently reaffirmed a Buy rating with an $85 price target, while Truist Financial has upgraded the stock to “Strong-Buy” and sees further upside on the back of pipeline momentum. Other firms tracked by Wall Street data providers cluster around a “Moderate Buy” consensus, reflecting growing conviction that the next leg of value will come from pipeline readouts rather than near-term CASGEVY sales.
How could CTX310 change cholesterol treatment?
Within the CRISPR Therapeutics Pipeline, CTX310 is the flagship cardiology program targeting patients with dangerous elevations in LDL cholesterol and triglycerides. The therapy edits the ANGPTL3 gene, aiming to inhibit the ANGPTL3 protein, a key regulator of lipid metabolism. High LDL and triglyceride levels are well-established drivers of coronary artery disease, a leading cause of death in the U.S. and a massive cost center for the healthcare system.
Today, high-risk patients often cycle through statins, PCSK9 inhibitors and other lipid-lowering drugs, frequently for life. By contrast, CTX310 is designed as a one-and-done in vivo gene-editing treatment that could permanently reset lipid levels after a single administration. CRISPR Therapeutics estimates that about 40 million people in the U.S. live with high triglyceride and LDL levels. The company will likely focus CTX310 on a much narrower, high-risk subset, but even a 0.1% addressable slice of that population — roughly 40,000 patients — would represent a substantial niche market for a premium, high-impact genetic therapy.
For investors, the appeal is clear: a durable, potentially curative product in a chronic-disease market that traditionally supports multi-billion-dollar annual sales for blockbuster drugs from companies like Apple-scale pharma peers. If CTX310 can demonstrate strong efficacy and a clean safety profile, it could help transform CRISPR Therapeutics from a single-product story into a diversified cardiovascular player competing, indirectly, with incumbent cardiometabolic franchises at giants comparable in scale to NVIDIA or Tesla within their own sectors.
Where does CTX320 fit in the CRISPR Therapeutics Pipeline?
Alongside CTX310, CTX320 is the second major cardiovascular asset in the CRISPR Therapeutics Pipeline. Instead of LDL and triglycerides, CTX320 targets lipoprotein(a), or Lp(a), an increasingly recognized independent risk factor for heart attacks and strokes. Elevated Lp(a) levels are largely genetically driven, and standard cholesterol drugs have limited impact on this marker, leaving many high-risk patients underserved.
CTX320 is also being developed as a potential one-time gene-editing treatment, opening the door to a functional cure for eligible patients with very high Lp(a). If successful, it would sit at the intersection of precision medicine and preventive cardiology, an area where no entrenched standard of care yet exists. That white space, combined with a relatively clear genetic target, helps explain why analysts at firms like Mizuho and Truist emphasize pipeline potential when defending bullish ratings despite current operating losses and minimal revenue.
Beyond cardiology, CRISPR Therapeutics is advancing additional programs, including an ex vivo cell therapy that could offer a functional cure for Type 1 diabetes. Together, these assets give the CRISPR Therapeutics Pipeline multiple shots on goal across large markets, even as the company continues to refine its platform and manufacturing capabilities.
How does CRISPR Therapeutics compare to other gene editors?
On NASDAQ, CRISPR Therapeutics trades alongside other gene-editing names and broader biotech peers tracked within the Medical – Biomedical and Genetics industry. While the stock has seen bouts of volatility and a recent pullback over the past month, several updates from financial platforms highlight how it has occasionally outperformed major benchmarks like the S&P 500 and Nasdaq Composite on single trading days when sentiment turns more optimistic.
Compared with large-cap innovators such as Apple-like leaders in their own domains, CRISPR Therapeutics remains a mid-cap speculative play: unprofitable, dependent on capital markets and partner funding, and highly sensitive to clinical and regulatory news flow. Yet the differentiated focus on one-time gene-editing approaches for common cardiometabolic risk factors sets it apart from many early-stage biotechs targeting ultrarare diseases only. For U.S. investors willing to accept heightened volatility, this positioning offers asymmetric upside if CTX310, CTX320 or the diabetes program deliver convincing data.
Still, the risks are substantial. Any serious safety issue in in vivo editing, lackluster efficacy, or unexpected regulatory hurdles could trigger a sharp re-rating. With revenue still limited and cash burn ongoing, setbacks in the CRISPR Therapeutics Pipeline would likely hit the share price much harder than in diversified pharma conglomerates.
In summary, the cardiology-focused CRISPR Therapeutics Pipeline — led by CTX310 and CTX320 — is increasingly central to how Wall Street values CRSP. If these programs evolve into first-in-class, one-time treatments for high-risk cardiovascular patients, the company could see a step-change in growth and investor perception. For U.S. portfolios seeking exposure to cutting-edge gene editing, the next data updates from these trials will be critical signposts for whether the risk-reward balance remains attractive.