Can Eli Lilly Foundayo and the Centessa deal push Lilly’s GLP-1 dominance into an even more powerful multi-drug growth story?

How big is the Eli Lilly Foundayo catalyst for Wall Street?
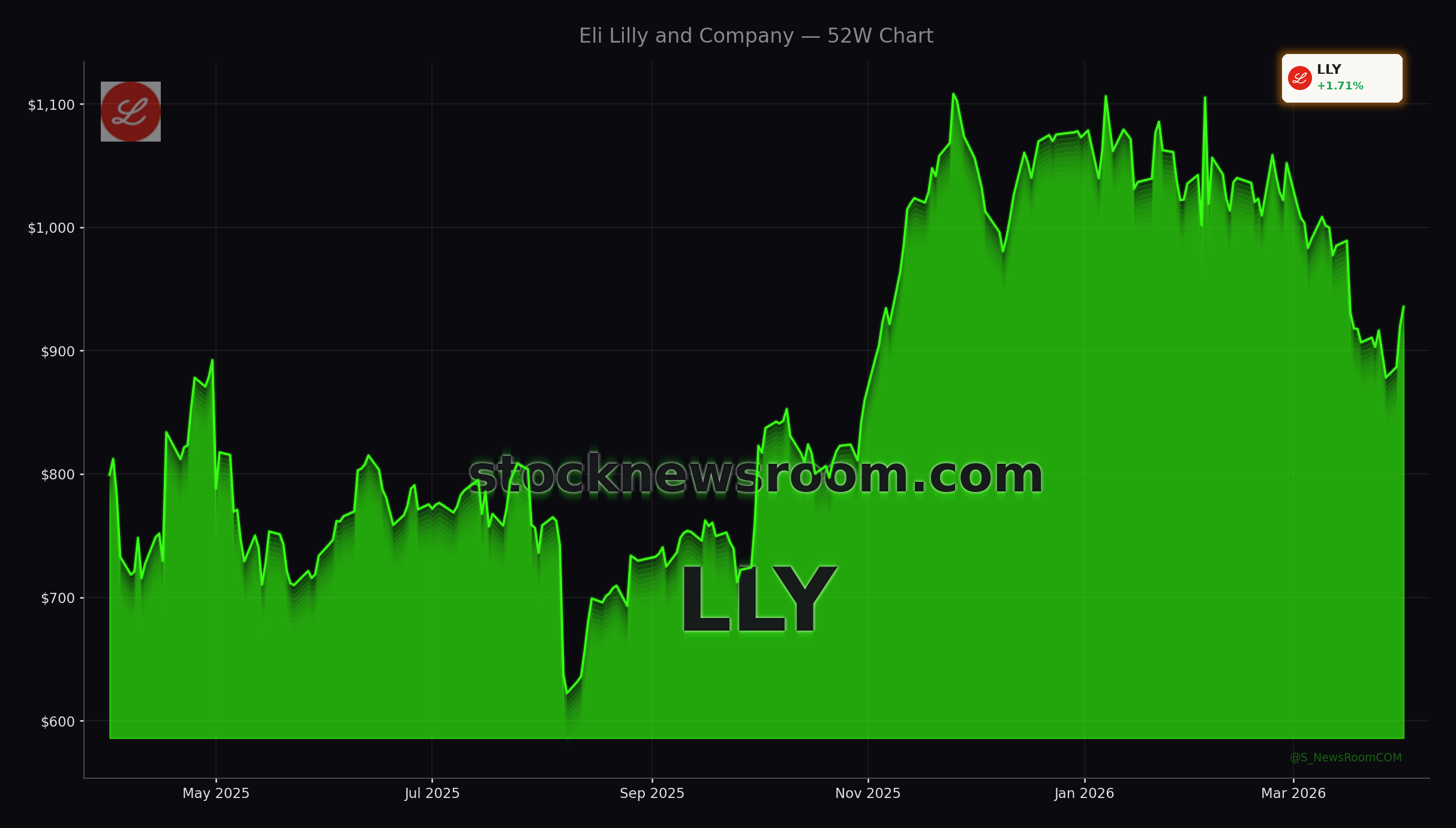
Eli Lilly and Company (LLY) traded sharply higher on Wednesday, rising to $961.90, a gain of about 4.58% versus Tuesday’s $920 close. The move keeps Lilly firmly among the most valuable healthcare names in the S&P 500 and adds fresh momentum to a stock that has already benefited from blockbuster demand for GLP-1 injectables Zepbound and Mounjaro.
Foundayo (orforglipron) is Lilly’s first oral GLP-1 therapy for chronic weight management and its second FDA-approved obesity medicine overall. Approved for adults with obesity, or overweight with weight-related medical problems, the once-daily pill is designed to be taken with or without food at any time of day, with no water or fasting restrictions. That simplicity differentiates it from Novo Nordisk’s oral Wegovy, which must be taken on an empty stomach with a waiting period before eating or drinking.
U.S. investors now have a clearer path to modeling a multi-product GLP-1 franchise that includes injectables and oral therapies, with Foundayo expected by many on Wall Street to be a multi-billion-dollar product over the coming decade.
What does Eli Lilly Foundayo mean for the GLP-1 pill race?
Clinical data from the ATTAIN-1 trial showed that patients on the highest dose of Foundayo who remained on treatment lost an average of 27.3 pounds, or 12.4% of body weight, over 72 weeks, compared with 0.9% for placebo. Across all participants, including those who discontinued treatment, average weight loss was 11.1% versus 2.1% for placebo. While Novo Nordisk’s oral Wegovy showed greater average weight loss of about 16.6% in its own studies, Foundayo’s flexible dosing and fewer restrictions could prove more attractive in everyday use.
Lilly is leaning heavily on that convenience message. Executives are highlighting that patients do not need to time the pill around meals or water intake, which could lower the real-world adherence barrier relative to competing regimens. For cost-sensitive U.S. patients, Foundayo will launch via LillyDirect with prices starting at $25 per month for commercially insured users with copay support, $149 for self-pay, and potential $50 pricing for some Medicare Part D patients later in 2026.
The Eli Lilly Foundayo launch also comes with substantial manufacturing backing. The company has pre-built roughly $1.5 billion in pill inventory and has committed over $55 billion to manufacturing expansion since 2020, giving it a scale advantage that could ease the supply bottlenecks that have plagued the first wave of weight-loss drugs.
How does the Centessa deal change Eli Lilly’s pipeline?
Alongside the Foundayo approval, Lilly agreed to acquire U.K.-based Centessa Pharmaceuticals for an upfront $6.3 billion, or $38 per share in cash, plus contingent value rights of up to $9 per share tied to future FDA approvals. The total potential value of the transaction is about $7.8 billion, with closing targeted for the third quarter of 2026, subject to shareholder and regulatory approvals. Major Centessa holders representing roughly 24% of shares have already backed the deal.
Centessa brings a new class of orexin receptor 2 agonists focused on sleep and wakefulness disorders. Lead candidate cleminorexton has shown promising Phase 2a data in narcolepsy and idiopathic hypersomnia, with the potential for a best-in-class profile. A second asset, ORX142, is also tied to the CVR milestones. For Lilly, the deal deepens its neuroscience platform and opens a strategic new vertical in sleep medicine that complements its metabolic and obesity portfolio.
The acquisition continues a dealmaking streak that includes recent agreements for Orna Therapeutics and Ventyx Biosciences, further diversifying revenue beyond GLP-1s while still leveraging Lilly’s expanding late-stage pipeline of 36 Phase 3 programs.
How strong is Lilly’s growth story versus peers?
Lilly’s GLP-1 franchise has already transformed its financial profile. In 2025, total revenue jumped roughly 45% to about $65.2 billion, while earnings per share surged 86% to $24.20. Management is guiding for 2026 revenue of $80 billion to $83 billion despite an expected low- to mid-teens pricing drag, underlining that volume demand for obesity and diabetes drugs remains extremely strong.
Mounjaro and Zepbound together generated more than $36 billion in 2025 revenue, with GLP-1 penetration in the U.S. obesity population still only in the mid-single digits. Foundayo adds another on-ramp for patients who dislike injections or do not perceive their weight as severe enough for injectable therapy, potentially broadening the addressable market again.
Compared with its primary rival Novo Nordisk, Lilly appears better positioned on manufacturing capacity and now has an answer to oral Wegovy. For U.S. investors used to looking at large-cap innovators like NVIDIA or Apple as secular growth stories, Lilly increasingly occupies a similar role in healthcare, with obesity, metabolic disease and now sleep medicine as its long-duration growth engines.
Speculation has also picked up that Lilly could follow companies such as Tesla and tech leaders with a stock split, given the share price now approaching $1,000. Commentators at The Motley Fool argue that surging GLP-1 revenues and a still-early penetration curve make a 2026 split plausible, though management has not announced any plans.
Related Coverage: What should investors read next?
For a deeper dive into how Lilly’s injectables and now Eli Lilly Foundayo fit into the broader GLP-1 landscape, including regulatory and concentration risks, readers can review Eli Lilly GLP-1 Strategy Boom and Revenue Shock. That analysis examines whether today’s explosive obesity-drug growth can remain a sustainable driver for LLY in diversified portfolios.
Investors following Big Pharma’s broader innovation race may also be interested in immunology developments. Our sector piece AbbVie Dermatology Data Boom at AAD 2026 Shocks Investors explores how AbbVie’s latest dermatology trial wins are shaping expectations for post-Humira growth, providing a useful counterpoint to Lilly’s obesity-led story.
As a convenient, once-daily oral pill that delivers meaningful weight loss, this is obesity care designed for the real world.— David A. Ricks, chair and CEO of Eli Lilly and Company
In summary, Eli Lilly Foundayo gives the company a powerful new lever in the booming obesity market just as the Centessa acquisition adds a high-upside sleep and neuroscience platform. For U.S. investors, Lilly now offers a rare combination of near-term revenue acceleration and longer-term pipeline optionality. The next few quarters of Foundayo prescriptions and regulatory progress on cleminorexton will show whether today’s optimism in the stock can translate into another leg of durable growth.