Can Eli Lilly Retatrutide’s powerful Phase 3 diabetes data reignite confidence in the GLP-1 boom despite rising competitive and pricing fears?

How market reacted to Eli Lilly’s new data?
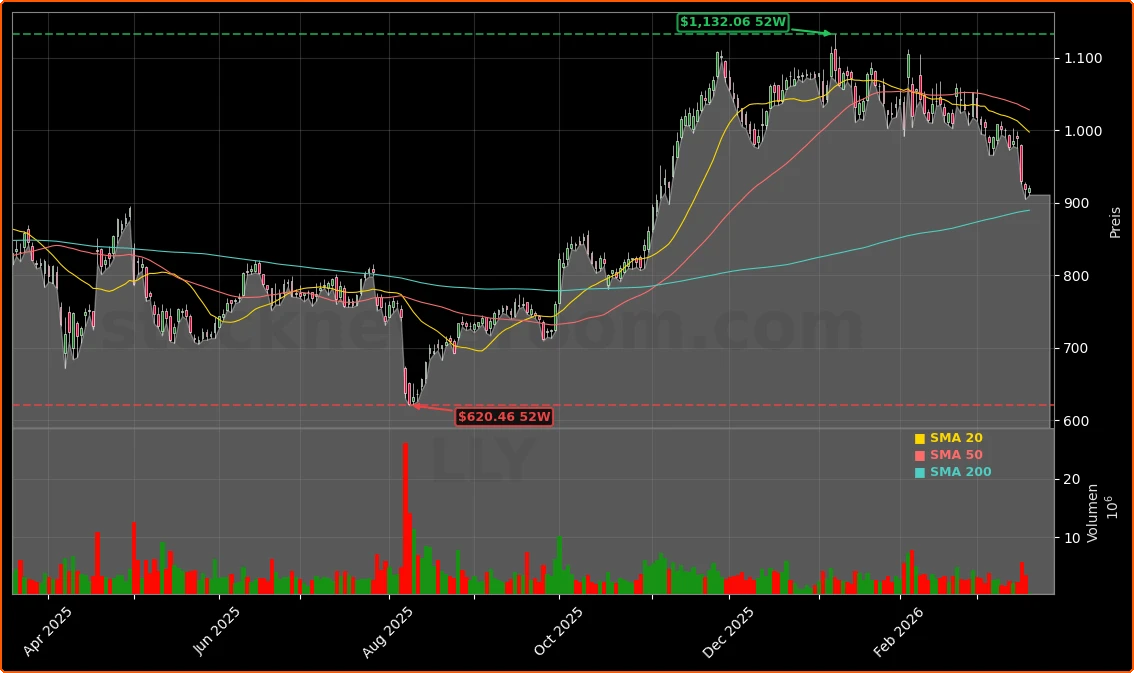
Despite the bullish clinical update, shares of Eli Lilly and Company (LLY) were little changed on Thursday, recently hovering around $919.80, up about 0.19% from the prior close. The stock has slid roughly 6% this week, coming under pressure as investors reassess long‑term GLP‑1 pricing power and factor in intensifying competition from Novo Nordisk and a wave of smaller biotechs. With Lilly still among the largest healthcare weights in the S&P 500, any shift in sentiment around its obesity and diabetes franchise ripples quickly through growth‑oriented portfolios on Wall Street and the NASDAQ.
Analyst conviction, however, remains broadly constructive. UBS this week reiterated a Buy rating and a $1,250 price target on Lilly, highlighting the company’s deep incretin pipeline, including Eli Lilly Retatrutide and the upcoming oral candidate orforglipron. That target implies meaningful upside from current levels, signaling that at least some large sell‑side firms see the recent pullback more as a consolidation in an overextended leader than the start of a structural derating.
What did the Eli Lilly Retatrutide trial show?
The TRANSCEND‑T2D‑1 Phase 3 study enrolled 537 adults with Type 2 diabetes inadequately controlled with diet and exercise alone and with no recent exposure to other diabetes drugs. Participants were randomized to once‑weekly retatrutide at 4 mg, 9 mg or 12 mg, or placebo, for 40 weeks. The primary endpoint was change in hemoglobin A1C, a key marker of long‑term blood sugar control.
At 40 weeks, Eli Lilly Retatrutide achieved average A1C reductions of 1.7% to 2.0% across the three doses using the efficacy estimand, versus a 0.8% reduction on placebo from a baseline of 7.9%. The highest 12 mg dose translated into nearly a full percentage point of incremental A1C benefit versus placebo, a clinically meaningful effect for patients early in their disease course. Importantly for long‑term morbidity, patients on the drug also showed improvements in several cardiovascular risk factors, including non‑HDL cholesterol, triglycerides and systolic blood pressure.
On the key secondary endpoint of weight, the data were even more striking. Participants on 12 mg lost an average of 36.6 pounds, or 16.8% of baseline body weight, using the efficacy estimand, compared with just 2.5% for placebo. Using the more conservative treatment‑regimen estimand that includes discontinuations and regimen changes, weight loss at the top dose still reached 15.3%, far ahead of placebo’s 2.6%. No plateau was observed through week 40, suggesting ongoing weight‑loss potential with longer treatment.
How does Eli Lilly Retatrutide stack up against rivals?
Eli Lilly is positioning retatrutide as a “triple G” next‑generation agent on top of its blockbuster tirzepatide franchise, which underpins Mounjaro for diabetes and Zepbound for obesity. Tirzepatide activates GLP‑1 and GIP receptors, whereas Eli Lilly Retatrutide adds glucagon receptor agonism, a combination that appears to further suppress appetite and enhance energy expenditure. In prior diabetes trials such as SURPASS‑1 and SURPASS‑2, tirzepatide achieved weight loss in the 11%–13% range at 40 weeks, below the 15%–17% seen so far with retatrutide in this Type 2 diabetes population.
There are no head‑to‑head studies yet against Novo Nordisk’s semaglutide‑based Wegovy and Ozempic portfolio, but the magnitude of weight reduction puts retatrutide toward the top of the current incretin class. At the same time, tirzepatide has delivered somewhat greater A1C reductions than those reported so far with retatrutide, underscoring that Lilly is building a toolbox of differentiated agents rather than a simple one‑drug franchise.
Beyond Novo Nordisk, investors are tracking competitive threats from emerging obesity players like Viking Therapeutics and Structure Therapeutics, as well as large‑cap pharma names such as Roche, which is expanding its AI‑driven R&D infrastructure with partners like NVIDIA. Yet Lilly’s ability to stack multiple late‑stage obesity and diabetes assets, combined with scale advantages in manufacturing, still makes it the reference name in the space for many U.S. growth managers.
Is safety a concern for Eli Lilly’s triple agonist?
The safety profile of Eli Lilly Retatrutide in TRANSCEND‑T2D‑1 looked broadly consistent with other injectable incretin therapies already on the market. The most common adverse events were gastrointestinal: nausea, diarrhea and vomiting, generally occurring during dose escalation. At the highest 12 mg dose, nausea affected about 26.5% of patients, diarrhea 22.8% and vomiting 17.6%, all above placebo but within ranges the market has come to expect for GLP‑1‑based drugs.
Discontinuation rates due to adverse events were relatively low at 2.2%, 4.5% and 5.1% across the 4 mg, 9 mg and 12 mg arms, respectively, with no such exits in the placebo group. A small share of patients reported dysesthesia, an unpleasant nerve sensation, which was generally mild and often resolved on treatment. For investors, the tolerability data help address one of the key risks around layering an additional glucagon pathway onto existing incretin mechanisms.
What’s next for Eli Lilly’s obesity and diabetes pipeline?
Eli Lilly plans to present full TRANSCEND‑T2D‑1 data at the American Diabetes Association Scientific Sessions in June and to publish detailed results in a peer‑reviewed journal. The company is running seven additional Phase 3 trials for retatrutide across obesity, Type 2 diabetes, obstructive sleep apnea, knee osteoarthritis, chronic low back pain, cardiovascular and renal outcomes, and metabolic dysfunction‑associated steatotic liver disease. Regulatory filings for obesity and diabetes indications are not expected until those readouts are further along, leaving some timeline uncertainty embedded in the current valuation.
In parallel, Lilly is advancing orforglipron, an oral GLP‑1 that could go before the FDA as early as the second quarter, positioning the company to challenge Novo Nordisk’s oral Wegovy in the burgeoning pill segment. Together with entrenched brands like Zepbound and Mounjaro, Eli Lilly Retatrutide strengthens a multi‑layered franchise that many on Wall Street view as one of the most powerful secular growth engines in global healthcare.
Related coverage: Investors weighing whether the latest data are enough to offset valuation and pricing concerns may also want to revisit our deeper look at recent sentiment shifts in Lilly. In Eli Lilly forecast warning after HSBC’s bearish reset, we examined how worries over GLP‑1 sustainability and competitive dynamics have sparked bouts of volatility in the name, despite its still‑dominant position in obesity and diabetes.
With triple agonist retatrutide, we set out to make a molecule that could help patients achieve substantial A1C reduction and weight loss.— Kenneth Custer, President, Lilly Cardiometabolic Health
Ultimately, the first Phase 3 win for Eli Lilly Retatrutide underscores how aggressively Lilly is extending its lead in metabolic disease beyond today’s GLP‑1 standards. For U.S. investors, the data support the long‑term growth story even as short‑term sentiment swings with every headline on pricing and competition. The next round of Phase 3 readouts and the ADA presentation will be crucial in determining whether Lilly’s triple‑agonist can become the next cornerstone of its obesity and diabetes portfolio.




