Can Eli Lilly’s new Foundayo pill and GLP‑1 pipeline keep its obesity lead as a brutal price war erupts?

How are Eli Lilly Weight Loss Drugs reshaping the stock story?
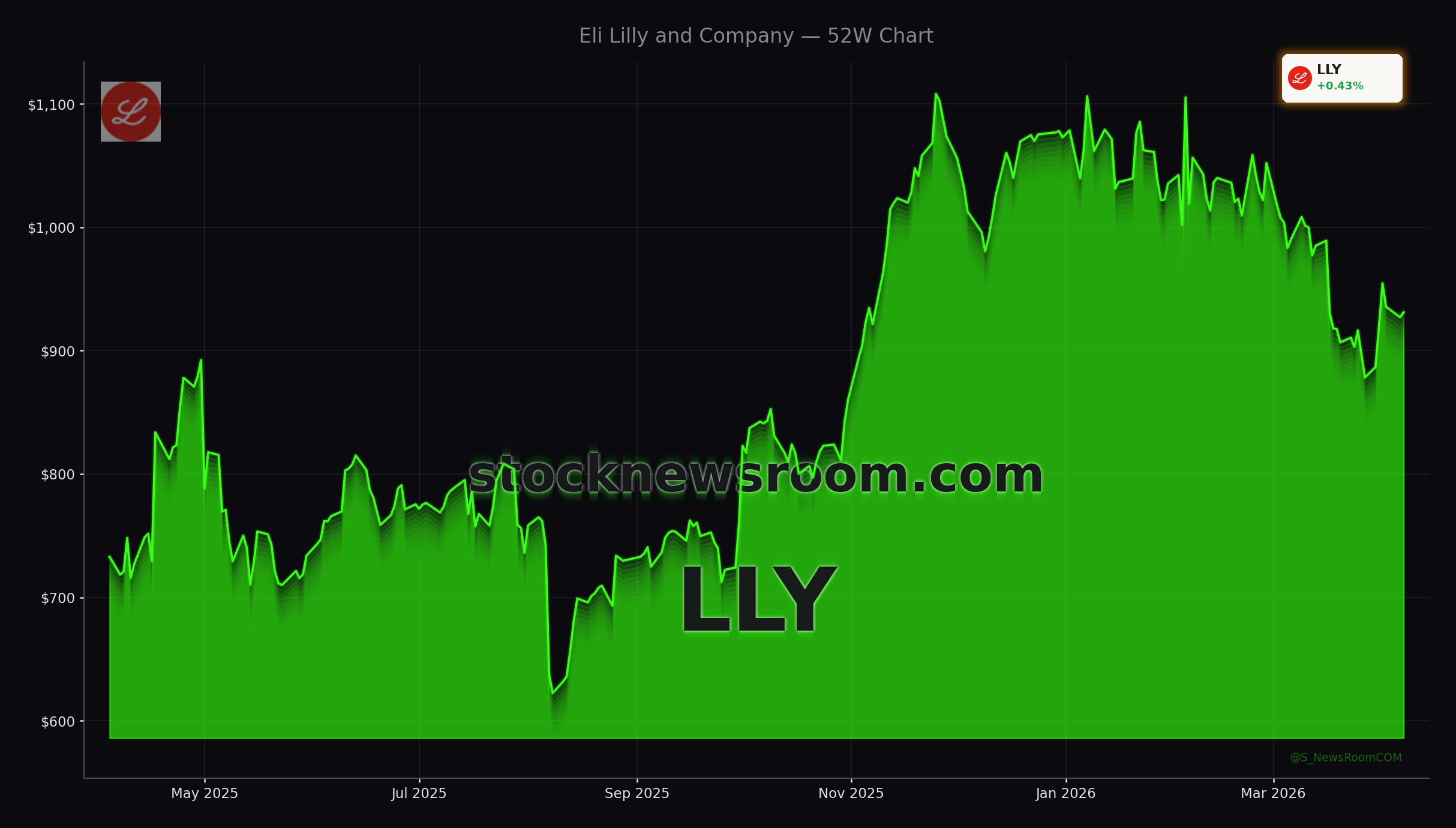
Eli Lilly and Company has quickly become the face of the global obesity drug boom, and the latest catalyst is Foundayo, a once‑daily oral GLP‑1 pill for weight loss approved by the FDA on April 1. The medicine is only the second oral anti‑obesity therapy to win U.S. clearance and stands out because it can be taken at any time of day without food or water restrictions, removing a key adherence hurdle compared with oral Wegovy. LLY shares, which have dropped roughly 13% so far in 2026, bounced after the approval but remain below recent peaks, leaving room for debate on whether the current valuation fully reflects the long‑term earnings power of Eli Lilly Weight Loss Drugs.
Foundayo is priced as low as $149 per month for the lowest dose for self‑pay patients, materially below the at least $299 per month that cash‑pay users face for comparable Zepbound doses. While its efficacy is lower than injectable Zepbound, management argues the pill will expand the addressable market by attracting patients who dislike injections or struggle with cold‑chain storage requirements. Wall Street firms such as Morgan Stanley and Goldman Sachs have reiterated bullish views on LLY in recent weeks, betting that incremental demand will outweigh any cannibalization of injection sales.
Can Foundayo offset Novo Nordisk’s Wegovy price offensive?
The approval lands just as Novo Nordisk escalates the GLP‑1 battle with a high‑dose version of Wegovy in the U.S. and a new cash‑pay price of about $399 per month, roughly 40% below what Eli Lilly charges for most Zepbound doses. Novo is also building out discounted subscription models to win back market share it has recently ceded to Lilly. That has ignited a price war in obesity drugs that threatens to compress margins industry‑wide and has already pressured shares of both companies.
For investors, the key question is whether the breadth of Eli Lilly Weight Loss Drugs can defend profits even as list prices adjust. Zepbound remains a best‑in‑class injectable with strong demand, and Foundayo opens a lower‑price, more convenient entry point that could help keep Lilly volumes high even if per‑patient revenue moderates. Analysts at JPMorgan and Bank of America continue to see Eli Lilly as the structural leader in the GLP‑1 space thanks to manufacturing scale and a broader pipeline than rivals such as Novo Nordisk and up‑and‑coming players like NVIDIA and Apple in the AI‑driven health data ecosystem, which are increasingly partnering across digital obesity and diabetes care.
What role does the GLP‑1 pipeline play beyond Foundayo?
Lilly is not standing still. Retatrutide, its weekly triple‑hormone receptor agonist, recently delivered impressive phase 3 data, cutting A1c by 1.72 percentage points and driving average weight loss of about 36.6 pounds while improving cardiovascular risk factors. The candidate is aimed at patients with severe obesity, including those with knee pain and high BMI who may not qualify for bariatric surgery or have failed current therapies. If successful, retatrutide could occupy a premium niche where payers may be more willing to reimburse higher prices because of reduced downstream healthcare costs.
Foundayo and retatrutide together underscore how Eli Lilly Weight Loss Drugs are being positioned across the severity spectrum, from oral entry‑level options to potent injectables for the most complex cases. On a recent investor call, CEO Dave Ricks said Lilly is prepared to “massively scale production” of Foundayo to meet expected global demand, with regulatory filings targeting more than 40 countries this year. The company is also investing in smart delivery and data platforms, overlapping with trends seen in diabetes technology markets where connected devices and analytics are becoming central to chronic‑disease management.
How important is the AC Immune Alzheimer’s collaboration?
While obesity dominates headlines, Lilly is quietly reinforcing its neurology franchise through an expanded collaboration with Swiss biotech AC Immune. The amended agreement extends work on Morphomer Tau small molecules designed to inhibit aggregation of the Tau protein, a core driver of neurodegeneration in Alzheimer’s disease and related disorders. AC Immune will receive an upfront payment of CHF 10 million and remains eligible for more than CHF 1.7 billion in future development, regulatory and commercial milestones, plus low double‑digit tiered royalties if products reach market.
The Tau candidates are optimized as brain‑penetrant oral small molecules that bind pathological conformations of Tau and have shown strong preclinical data in halting aggregation and seeding across disease stages. IND‑enabling studies are expected to start in the first half of 2026. For Lilly, this deal expands an already significant Alzheimer’s pipeline and diversifies the long‑term revenue base beyond Eli Lilly Weight Loss Drugs, helping reassure investors worried about over‑reliance on GLP‑1 economics.
Related Coverage
For a deeper dive into how pricing, AI‑driven manufacturing and competitive dynamics intersect in obesity care, readers can explore an in‑depth look at Eli Lilly’s broader weight‑loss strategy versus Novo Nordisk, which examines whether the company can maintain its edge as the price war intensifies. On the competitive side, a detailed analysis of Novo Nordisk’s Wegovy high‑dose rollout explains how the Danish rival aims to reclaim share in the GLP‑1 arena and what that could mean for Lilly’s growth trajectory over the next few years.
Overall, the expanding portfolio of Eli Lilly Weight Loss Drugs, from Zepbound to the newly approved Foundayo and late‑stage candidate retatrutide, keeps the company at the center of the global obesity market despite mounting price competition from Novo Nordisk. The reinforced Alzheimer’s partnership with AC Immune adds another long‑duration growth pillar, giving LLY a broader innovation story than GLP‑1s alone. For long‑term investors, the next key milestones will be Foundayo’s U.S. launch metrics, retatrutide’s regulatory path and early data from Morphomer Tau programs, all of which will help determine how much upside remains in the stock as the obesity drug race evolves.