Can the Eli Lilly Weight-Loss Strategy stay on top as Novo Nordisk slashes prices and AI reshapes the obesity drug race?

Can Eli Lilly Weight-Loss Strategy hold its market lead?
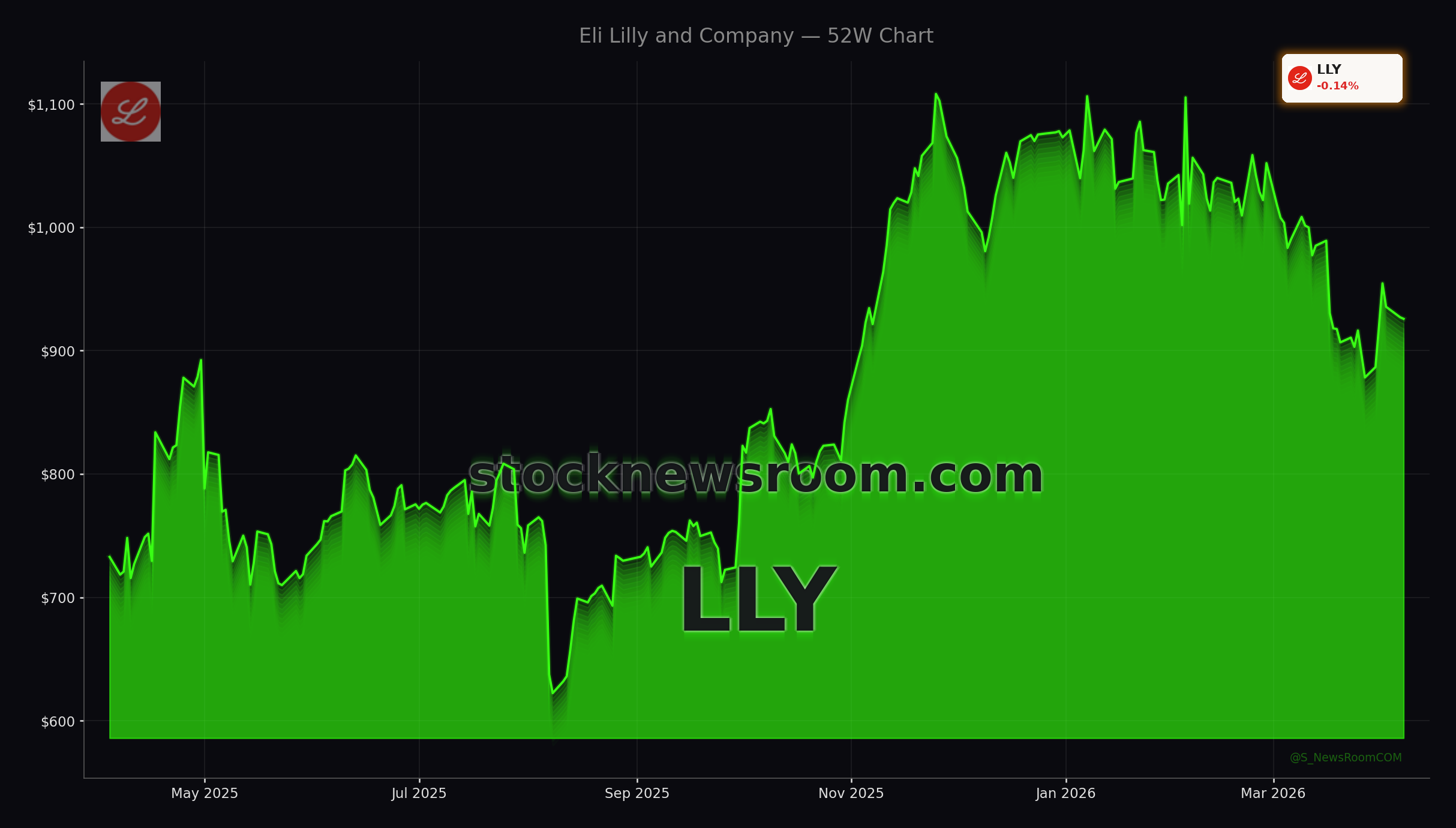
Eli Lilly has become the flagship obesity play on the NYSE thanks to injectable GLP-1/GIP drug Zepbound and now oral GLP-1 Foundayo, approved by the FDA on April 1 for weight loss and weight-related conditions. Shares have fallen about 13% so far in 2026, but a recent bounce after the Foundayo green light has revived the bull case that Lilly can defend and even extend its lead in the global GLP-1 market.
The Eli Lilly Weight-Loss Strategy is now clearly multi-pronged. Zepbound remains the efficacy benchmark in injections, while Foundayo targets a new population of patients who have avoided needles or struggled with storage requirements for injectables. Foundayo is only the second oral anti-obesity drug approved in the U.S., and unlike oral Wegovy from rival Novo Nordisk, it can be taken without food or water timing restrictions, a convenience edge that could matter in primary care adoption.
Importantly for payers and patients, Foundayo’s list price for the lowest strength starts as low as $149 per month for self-pay, roughly half the minimum $299 per month that Zepbound users without insurance must pay. Even if efficacy is lower than Zepbound, management is betting Foundayo will expand the overall GLP-1 market rather than simply cannibalize existing injectable revenue.
How tough is Novo Nordisk’s pricing and efficacy challenge?
Novo Nordisk has not ceded the obesity crown without a fight. In the U.S., the Danish drugmaker has launched a high-dose version of Wegovy, responding directly to share losses in obesity to Lilly’s rapidly scaling GLP-1 franchise. Novo has also set a cash price of roughly $399 per month for its new high-dose “WeGovy shot”, estimated to be about 40% cheaper than Lilly’s most commonly used Zepbound doses for self-pay patients.
On the oral side, Novo’s strategy is equally aggressive. Oral Wegovy has hit the market with explosive initial demand at a discount entry price for self-pay patients reportedly below $4 per day in some promo structures, undercutting Lilly’s injectable options and aiming to draw in needle-averse users. A recent Novo analysis indicated oral Wegovy delivered about 3.2 percentage points more weight loss than Lilly’s Foundayo in cross-trial comparisons, while Foundayo saw higher dropout rates from side effects. That gives Novo a compelling efficacy narrative even as Lilly touts Foundayo’s dosing flexibility.
The net result is an intensifying tug-of-war that could erode the outsized obesity margins that drove both stocks to massive gains in 2023–2024. Analysts at MarketBeat highlight that Wall Street Zen recently downgraded Lilly from “Strong Buy” to “Buy,” even as the broader analyst community maintains a “Moderate Buy” stance with an average target around $1,222, implying meaningful upside from current levels if the Eli Lilly Weight-Loss Strategy remains dominant.
Where do AI and NVIDIA fit into Eli Lilly Weight-Loss Strategy?
Lilly is not relying on GLP-1s alone. The company has signed a multiyear, multibillion-dollar collaboration with NVIDIA to build out AI-driven drug discovery and development capabilities. Over five years, the partners plan to use advanced GPU computing and simulation to accelerate pipeline design, optimize clinical trial protocols, and identify novel combinations of hormones and pathways that could create the next generation of obesity and metabolic drugs beyond today’s GLP-1s and GLP-1/GIP agonists.
For investors, this AI pact is a long-dated call option: near-term, it will add to R&D and cloud spending; longer term, it could shorten development timelines and reduce late-stage failures, potentially boosting returns on capital across Lilly’s portfolio. The AI work is highly relevant for the Eli Lilly Weight-Loss Strategy, as the company pushes beyond Zepbound and Foundayo into higher efficacy or more tolerable agents.
One standout candidate is retatrutide, a weekly triple-hormone receptor agonist that recently generated impressive Phase 3 data, including average weight loss of about 36.6 pounds and meaningful A1c reduction of 1.72 percentage points, alongside improvements in cardiovascular risk factors and knee pain. Retatrutide is being positioned for patients with very high BMI who might otherwise be candidates for bariatric surgery, giving Lilly a potential foothold in the most severe segment of the obesity market.
How do neurology deals broaden Lilly beyond GLP-1s?
Parallel to its obesity push, Lilly is deepening its pipeline in neurodegenerative diseases, which could diversify revenue away from GLP-1 concentration risk. Swiss biotech AC Immune has amended its 2018 Morphomer Tau license and collaboration with Lilly, focused on small molecules targeting intracellular Tau aggregation for Alzheimer’s and related disorders. The new deal extends work on next-generation Tau Morphomer candidates and backups, with Investigational New Drug (IND)-enabling studies slated for the first half of 2026.
AC Immune will receive a CHF 10 million upfront payment and a Phase 1 initiation milestone, on top of previously agreed potential milestones exceeding CHF 1.7 billion plus low double-digit royalties if products reach market. For Lilly, the partnership complements its existing Alzheimer’s efforts and underscores a broader strategy: use today’s GLP-1 cash flows to fund high-risk, high-reward R&D bets in neurology and beyond.
Related coverage: GLP-1 war and portfolio angles
Investors who want a deeper dive into Foundayo’s launch and its role in multi-drug growth can review detailed analysis of Lilly’s GLP-1 pill rally and Centessa acquisition strategy, which explores how multiple obesity assets might compound long-term revenue. On the competitive front, Novo’s response is examined in coverage of Novo Nordisk’s Wegovy high-dose strategy, outlining how aggressive dosing and pricing tactics aim to claw back share from Lilly’s GLP-1 franchise and what that could mean for margins across the sector.
In summary, the Eli Lilly Weight-Loss Strategy now spans injectables, oral pills, next-wave triple agonists, AI discovery with NVIDIA, and diversification into neurology—all while navigating a fierce pricing and efficacy battle with Novo Nordisk. For U.S. investors, Lilly remains a high-conviction but higher-volatility large-cap growth play, where execution on Foundayo and retatrutide will be critical to justify premium valuation. The next catalysts around retatrutide data, AI updates, and the AC Immune Tau program will show whether the Eli Lilly Weight-Loss Strategy can keep powering long-term returns despite mounting competition.