Can Johnson & Johnson’s FDA approval of ICOTYDE really shift the psoriasis market enough to move a $575 billion healthcare giant?

How does Johnson & Johnson FDA approval ICOTYDE move the stock?
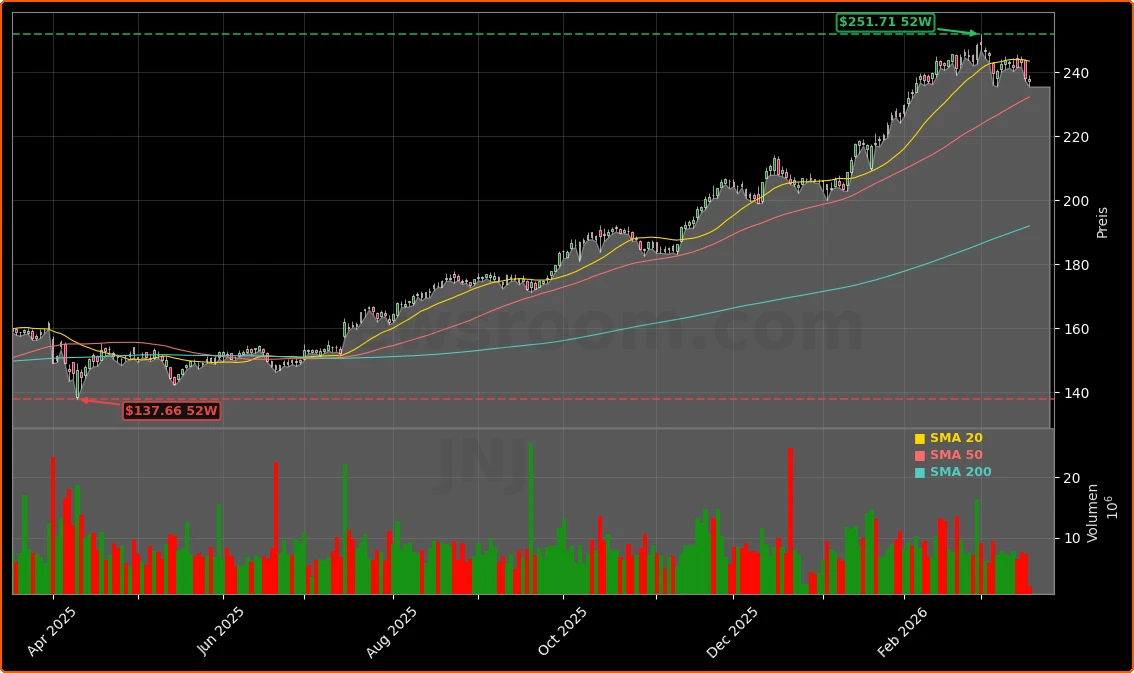
Johnson & Johnson shares recently traded around $236.97, off about 0.5% from the prior close of $238.70 and below the 52‑week high of $251.71, suggesting the Johnson & Johnson FDA approval ICOTYDE was at least partly priced in after a strong 12‑month rally. The company’s roughly $575 billion market capitalization means even a sizable new drug launch will take time to visibly shift consolidated revenue, but the psoriasis pill strengthens an already robust Innovative Medicine growth narrative.
Citigroup analysts model peak ICOTYDE sales of about $5.5 billion, arguing that the drug delivers biologic‑level efficacy with the convenience of a once‑daily oral pill – a combination that has eluded previous psoriasis therapies. In a global psoriasis market measured in the tens of billions of dollars annually, such a profile positions Johnson & Johnson to capture share from injectable biologics and competing oral agents, potentially supporting upside to long‑term revenue and earnings forecasts.
On Wall Street, JNJ already benefits from a broadly positive backdrop. HSBC recently raised its price target to $280, citing oncology momentum and the stock’s defensive appeal, while other research houses continue to rate the name a buy or moderate buy. For U.S. investors comparing large‑cap healthcare peers like Apple‑adjacent digital health players or diversified pharmas such as Merck and AbbVie, ICOTYDE adds another distinct growth pillar to JNJ’s investment case.
What makes ICOTYDE clinically different?
ICOTYDE is an interleukin‑23 (IL‑23) receptor antagonist approved for adults and pediatric patients 12 years and older who weigh at least 40 kg and are candidates for systemic therapy or phototherapy. It is the first and only targeted oral peptide that precisely blocks the IL‑23 receptor, a key driver of the inflammatory cascade in psoriasis. Patients take one 200 mg tablet once daily on an empty stomach with water, at least 30 minutes before food.
The approval rests on the ICONIC Phase 3 program, which enrolled about 2,500 patients across multiple trials in adults and adolescents and in high‑impact sites such as scalp and genital psoriasis. In head‑to‑head superiority studies against an active comparator, roughly 70% of patients achieved clear or almost clear skin (Investigator Global Assessment 0/1) and 55% reached PASI 90 at Week 16. Adverse reactions through Week 16 were within 1.1% of placebo, and no new safety signals emerged through Week 52, reinforcing the narrative of a favorable risk‑benefit profile.
From a safety standpoint, prescribers must monitor for infections and screen for tuberculosis before starting therapy, in line with other immune‑modulating agents. Common side effects include headache, nausea, cough, fatigue and fungal infections. Still, Johnson & Johnson FDA approval ICOTYDE underscores the FDA’s comfort with the overall benefit‑risk balance, especially given the long‑standing unmet need for convenient, effective oral options.
How big is the commercial opportunity for Johnson & Johnson?
Plaque psoriasis affects more than 8 million people in the U.S. and over 125 million worldwide, with around one‑quarter of cases classified as moderate‑to‑severe. Many patients cycle repeatedly through topical therapies before moving to systemic treatment, even as updated guidance from the International Psoriasis Council recommends earlier escalation when topicals fail. ICOTYDE’s once‑daily oral dosing, combined with strong efficacy, directly targets this transition point.
For Johnson & Johnson, the franchise extends beyond skin disease. ICOTYDE is being studied in psoriatic arthritis, ulcerative colitis and Crohn’s disease, opening the door to a broader immunology platform if future trials succeed. The drug originated from a collaboration with Protagonist Therapeutics, which now receives a $50 million milestone payment and could earn up to $580 million in additional milestones plus tiered royalties, while JNJ retains global commercial rights.
Strategically, the Johnson & Johnson FDA approval ICOTYDE helps offset looming biosimilar pressure on legacy brands such as STELARA and supports the company’s effort to keep its pharma segment growing above the broader market. It also strengthens JNJ’s position versus psoriasis competitors, including AbbVie, Eli Lilly and other big‑pharma players followed closely by investors in high‑growth names like Tesla and NVIDIA looking for diversified exposure.
ICOTYDE delivers something unique in psoriasis treatment – combining skin clearance with a favorable safety profile in a once‑daily pill.— Linda Stein Gold, M.D., Director of Dermatology Clinical Research, Henry Ford Health
In summary, Johnson & Johnson FDA approval ICOTYDE marks a pivotal milestone for the company’s immunology pipeline, delivering a first‑in‑class oral IL‑23 receptor therapy that could become a multi‑billion‑dollar asset. For investors, the launch reinforces JNJ’s profile as a defensive S&P 500 stalwart with credible growth optionality, and upcoming readouts in psoriatic arthritis and inflammatory bowel disease will be key catalysts to watch over the next few years.