Does Moderna’s billion‑dollar patent peace deal finally clear the runway for its mRNA pipeline—or just cap the upside?
Moderna Patent Settlement: Why Wall Street Is Cheering the Move
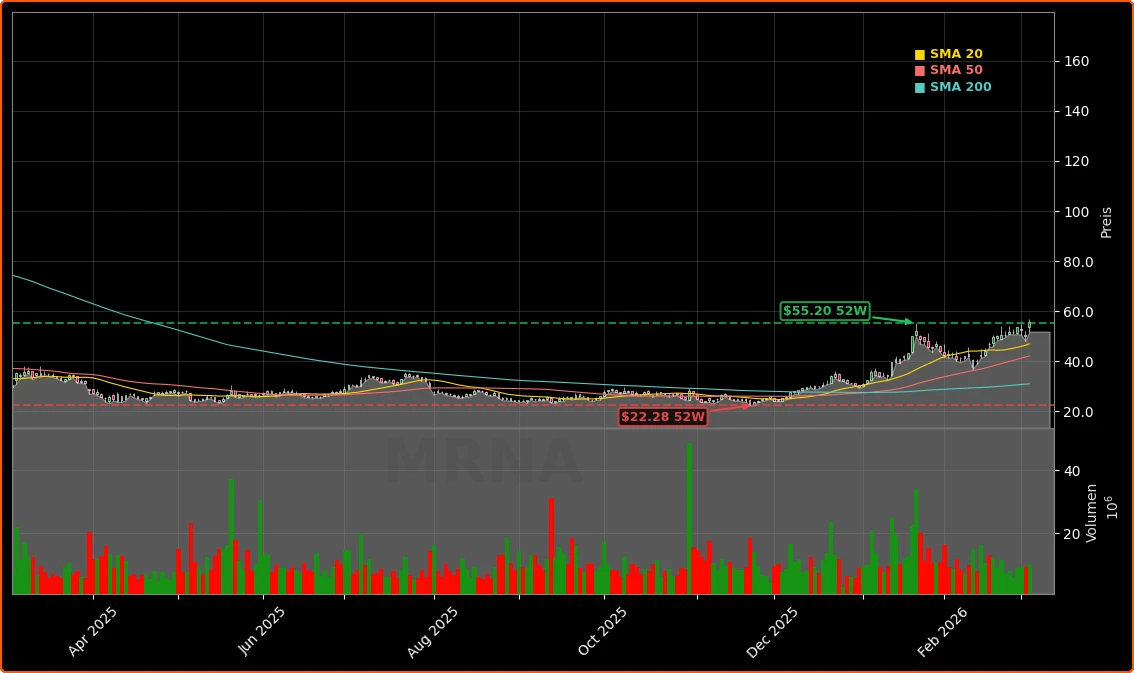
Moderna shares surged more than 10% in pre‑market trading and held an intraday gain of about 11% around $55.47 on Wednesday after the company unveiled a global settlement with Arbutus Biopharma and Genevant Sciences. The agreement ends years of patent litigation over the lipid nanoparticle (LNP) delivery technology underlying Moderna’s COVID‑19 vaccine Spikevax and its RSV vaccine mRESVIA. For investors, the Moderna Patent Settlement removes a hard‑to‑quantify legal threat that had been depressing valuation multiples, even as the company pushed forward with a broader mRNA pipeline.
Under the deal, Moderna will pay $950 million in July 2026 and could pay an additional $1.3 billion depending on the outcome of an appeal related to U.S. government‑contractor immunity under Section 1498. Crucially, the settlement wipes out all current and future royalty claims on Spikevax, mRESVIA and a suite of infectious‑disease candidates that use the same LNP platform. That gives the market a much clearer line of sight on Moderna’s future gross margins and removes the risk of an open‑ended royalty stream or a surprise trial verdict.
Wall Street’s relief is visible not just in the price action but also in the tone of commentary. The consensus is that while a potential $2.25 billion payout is far from ideal, it is a manageable outcome given Moderna’s remaining cash and the strategic value of full freedom to operate on its core technology stack. With the Moderna Patent Settlement now largely quantified, the stock can start trading more on pipeline progress and less on courtroom headlines.
Moderna, Inc.: What Exactly Is in the Patent Deal?
The settlement structure matters for valuation. Moderna will book a $950 million charge in Q1 2026 tied to the upfront payment due in the third quarter of 2026. That upfront resolves all worldwide litigation concerning Spikevax and mRESVIA and covers Moderna’s wider infectious‑disease portfolio, including combination candidates like mRNA‑based flu/COVID vaccines (such as its mNEXSPIKE and mCOMBRIAX programs). There are no ongoing royalties under this agreement, which is a key win given the potentially long commercial life of these products.
The additional $1.3 billion is contingent. Moderna is appealing to the U.S. Court of Appeals for the Federal Circuit, arguing that as a government contractor during the pandemic it should enjoy broad immunity from patent damages under Section 1498. If the company prevails, that contingent payment disappears entirely. If it loses, Moderna would have to pay the additional sum within a defined window (roughly within 90 days of a negative ruling), subject to the precise terms of the final judgment. Management has signaled it does not currently view a full loss on appeal as the most likely outcome and is not recording a reserve for the $1.3 billion piece.
In exchange, Moderna gains a worldwide, non‑exclusive license to the Arbutus/Genevant LNP technology for SM‑102‑containing mRNA vaccines directed at infectious diseases, plus a covenant not to sue for certain patents and products. The company has also consented to a court judgment of infringement and no invalidity on four asserted patents. From an investor standpoint, that legal concession is far less important than the practical result: Moderna’s existing vaccine franchises and much of its pipeline are now protected by a clear, royalty‑free license rather than left dangling over future lawsuits.
Moderna, Inc.: Balance Sheet Impact and Liquidity After the Settlement
Investors naturally worry when a mid‑cap biotech takes on potential multi‑billion‑dollar obligations. Here, the numbers are important. Moderna expects to end 2026 with $4.5 billion to $5.0 billion in cash and cash equivalents even under a scenario where it pays the full $2.25 billion. That is lower than a prior cash outlook of roughly $5.5 billion to $6.0 billion, but still leaves substantial financial flexibility. On top of balance‑sheet cash, Moderna also has access to a $900 million credit facility, bringing potential year‑end 2026 liquidity to $5.4 billion to $5.9 billion.
The initial $950 million charge will depress reported earnings in 2026, but this is a one‑time non‑cash accounting impact in the near term and a finite cash drain in the medium term. The absence of future royalties can, over time, offset part of that burden if Spikevax, mRESVIA and upcoming combo vaccines achieve meaningful commercial traction. Importantly, Moderna reiterates that it expects revenue growth to return in 2026 and still targets breakeven by 2028 as its portfolio diversifies beyond COVID.
Citigroup has weighed in, arguing that while a potential $2.3 billion hit (counting the contingent piece) is clearly not the best‑case outcome, it is also not the nightmare scenario some feared. The firm maintains a “Neutral” rating on Moderna with a $41 price target, which is well below the current $55+ share price. That underscores an important nuance: the Moderna Patent Settlement may have removed a major downside risk, but some analysts still see the stock as fully valued or stretched relative to lingering demand uncertainty for COVID boosters and execution risk in the broader pipeline.
Moderna, Inc.: Pipeline, Growth Outlook and Key Catalysts
With the patent cloud lifted, management is emphasizing growth drivers rather than litigation recovery. The company expects to return to year‑over‑year revenue growth in 2026, driven primarily by respiratory vaccines. It is guiding for regulatory decisions this year on its flu‑only and flu/COVID combination vaccines, which could create a more predictable seasonal revenue base starting in 2026–2027. That transition from a pandemic‑driven one‑off windfall to a recurring, albeit smaller, endemic revenue stream is central to the Moderna bull case.
Beyond respiratory, Moderna is leaning heavily on oncology and rare diseases to rebuild its growth narrative. A key asset is its personalized cancer vaccine program (mRNA‑4157) in combination with Merck’s Keytruda for melanoma and potentially other solid tumors. Early‑stage data have shown meaningful reductions in recurrence and death compared with Keytruda alone, and Phase 2 and Phase 3 programs are now underway. If successful, this could be the first broadly commercialized personalized mRNA cancer vaccine, opening the door to multi‑billion‑dollar long‑term revenue.
That kind of pipeline optionality is why some growth‑oriented investors lump Moderna together with other high‑beta innovators such as NVIDIA in AI chips or Tesla in electric vehicles: once a platform proves itself, incremental products can scale quickly off a shared R&D and manufacturing base. In Moderna’s case, the Moderna Patent Settlement is effectively a platform‑protection event, clarifying that much of the infectious‑disease side of its technology stack is now de‑risked from a legal standpoint. Over the next 12–24 months, the key catalysts will be regulatory approvals for respiratory vaccines, updated commercial guidance on endemic COVID demand, and interim or full data readouts from oncology and rare‑disease trials.
Moderna, Inc.: Competitive Landscape Versus Big Vaccine Players
Investors must consider where Moderna fits in a post‑pandemic vaccine market dominated by giants like Pfizer and Johnson & Johnson, with mRNA rival BioNTech as another critical reference point. While Moderna is not yet a member of the S&P 500’s mega‑cap elite like Apple, it has a unique position as a pure‑play mRNA platform company with a significant cash war chest. The Moderna Patent Settlement, by removing royalty obligations and litigation risk around LNP technology, arguably strengthens its hand versus peers.
Moderna continues to pursue litigation of its own against Pfizer and BioNTech over alleged infringement of Moderna’s mRNA vaccine patents. Management has argued that, on a net basis, the value of its patent assets across proceedings exceeds the liabilities such as those in the Arbutus/Genevant matter. That claim has not yet been fully tested in court, but it suggests Moderna views its intellectual‑property portfolio as a strategic weapon rather than just a defensive moat. For U.S. investors, this raises the possibility of additional settlement income or royalty streams down the line, even as the company writes a large check in the current case.
On the commercial front, Moderna’s challenge will be to stabilize COVID revenue in a competitive, price‑sensitive booster market while expanding into adjacent segments such as RSV and flu. Larger, diversified pharmas can cross‑subsidize vaccine launches from other business lines, whereas Moderna remains heavily dependent on the success of a handful of products. That concentration risk is one reason some analysts, including Citigroup, remain cautious despite the positive news flow around the Moderna Patent Settlement.
Still, for long‑term growth investors comfortable with volatility, Moderna’s situation resembles that of earlier‑stage disruptors like NVIDIA before its AI boom: a proven core technology, a fortified balance sheet, and a pipeline of potential blockbusters. The key difference is that healthcare regulation and payer dynamics create longer, more uncertain commercialization timelines than in consumer tech or autos, where companies like Tesla operate. That makes careful position sizing and a multi‑year time horizon essential for anyone considering an allocation.
Moderna, Inc.: Valuation, Risk/Reward and Portfolio Role
At around $55 per share, Moderna has rebounded sharply from its lows but still trades well below the euphoric peaks of the pandemic era. Year‑to‑date performance has been strong, with some commentary highlighting gains of more than 70% before this latest jump. The Moderna Patent Settlement has clearly helped re‑rate the stock by removing a tail‑risk scenario in which open‑ended damages or ongoing royalties would have materially impaired long‑term free cash flow.
Even after Wednesday’s move, Moderna is not priced like a mature, stable pharma; it is still valued as a volatile growth asset whose future depends on execution in multiple high‑risk clinical programs. Bulls argue that the current price understates the potential of its oncology pipeline and underappreciates the durability of respiratory vaccine revenues as COVID becomes an endemic booster market similar to flu. Bears counter that post‑pandemic vaccine demand could disappoint, that oncology timelines may slip, and that further dilution or higher leverage could be needed if cash burn persists longer than expected.
Conclusion
From a portfolio‑construction standpoint, Moderna makes the most sense as a satellite position within a diversified healthcare or growth‑equity allocation, rather than as a core S&P 500 proxy. Its beta is high, its earnings visibility is limited, and binary trial outcomes can move the stock dramatically in either direction. However, the Moderna Patent Settlement reduces one major non‑fundamental risk (adverse litigation shock), making the risk/reward profile cleaner: investors are now betting more on science, execution and market adoption than on courtroom surprises.
Further Reading
- Moderna, Inc. (MRNA) Stock Price, News, Quote & History (Yahoo Finance)
- Moderna rises as up to $2.25 billion settlement for COVID vaccine patent dispute to remove overhang (Reuters)
- Why Moderna’s $950M Patent Bill Is A Small Price For Pipeline Freedom (Seeking Alpha)
- Moderna and Ross Stores Lead Stock Gainers on Wednesday (24/7 Wall St.)