Can the Pfizer Lyme vaccine’s >70% efficacy data revive sentiment around PFE just as investors write off its post-COVID growth story?

How strong are the VALOR efficacy results?
Pfizer Inc. and Valneva said their investigational 6-valent OspA-based Lyme disease vaccine candidate, PF‑07307405 (also known as LB6V), achieved more than 70% efficacy in preventing confirmed Lyme disease cases in the Phase 3 VALOR trial. In prespecified analyses for the second Lyme season, efficacy reached 73.2% from 28 days after the fourth dose and 74.8% from one day after dose four versus placebo. Participants aged five years and older received a four-dose regimen spread over roughly one year, targeting regions in the U.S., Canada, and Europe with high Lyme incidence.
The trial did enroll fewer Lyme cases than expected, and the primary statistical criterion – a 95% confidence interval lower bound above 20% in the first prespecified analysis – was not met. However, that threshold was exceeded in the second prespecified analysis, and Pfizer emphasized that the overall efficacy profile is clinically meaningful. The candidate was described as well tolerated with no new safety concerns identified at the time of analysis, an important factor for vaccines intended for broad, mostly healthy populations.
Why does a Pfizer Lyme vaccine matter to investors?
A successful Pfizer Lyme vaccine could open up a sizeable niche market in North America and Europe, where Lyme disease is the most common tick-borne infection and diagnoses have climbed steadily over the last decade. The U.S. Centers for Disease Control and Prevention estimates roughly 476,000 Americans are diagnosed and treated for Lyme disease annually, while European surveillance systems report over 100,000 cases per year. There is currently no approved human Lyme vaccine, making LB6V the most advanced program globally and positioning Pfizer to define the category if regulators sign off.
From a portfolio perspective, the Lyme shot would complement Pfizer’s existing vaccine franchise, which includes pneumococcal, meningococcal, and RSV products. As COVID-19 vaccine revenues normalize, management has been under pressure to show that late-stage pipeline assets can restore growth. A green light for the Pfizer Lyme vaccine over the next couple of years would diversify vaccine cash flows and help offset patent cliffs in other therapeutic areas, including oncology and immunology.
What are the next steps for Pfizer and Valneva?
On the back of the VALOR data, Pfizer plans to submit regulatory applications in key markets, likely starting with the U.S. Food and Drug Administration and European regulators. Exact timelines have not been disclosed, but late-stage data at this level of detail typically precede formal filings by several months. Under their collaboration agreement, Pfizer will take the lead on manufacturing and commercialization if the Pfizer Lyme vaccine is approved, while Valneva participates through milestones and royalties.
Regulators will scrutinize the mixed statistical picture – particularly the missed primary VALOR criterion – alongside the strong second analysis and clean safety profile. Advisory committee discussions could focus on disease burden, risk–benefit in children, and how to position the vaccine in relation to current prevention strategies such as tick avoidance and early antibiotic treatment. If successful, the product could eventually be recommended for people living in or traveling to high-risk regions, giving it a profile somewhat similar to certain travel and seasonal vaccines already marketed by Valneva and other players.
How does this fit into Pfizer’s stock story?
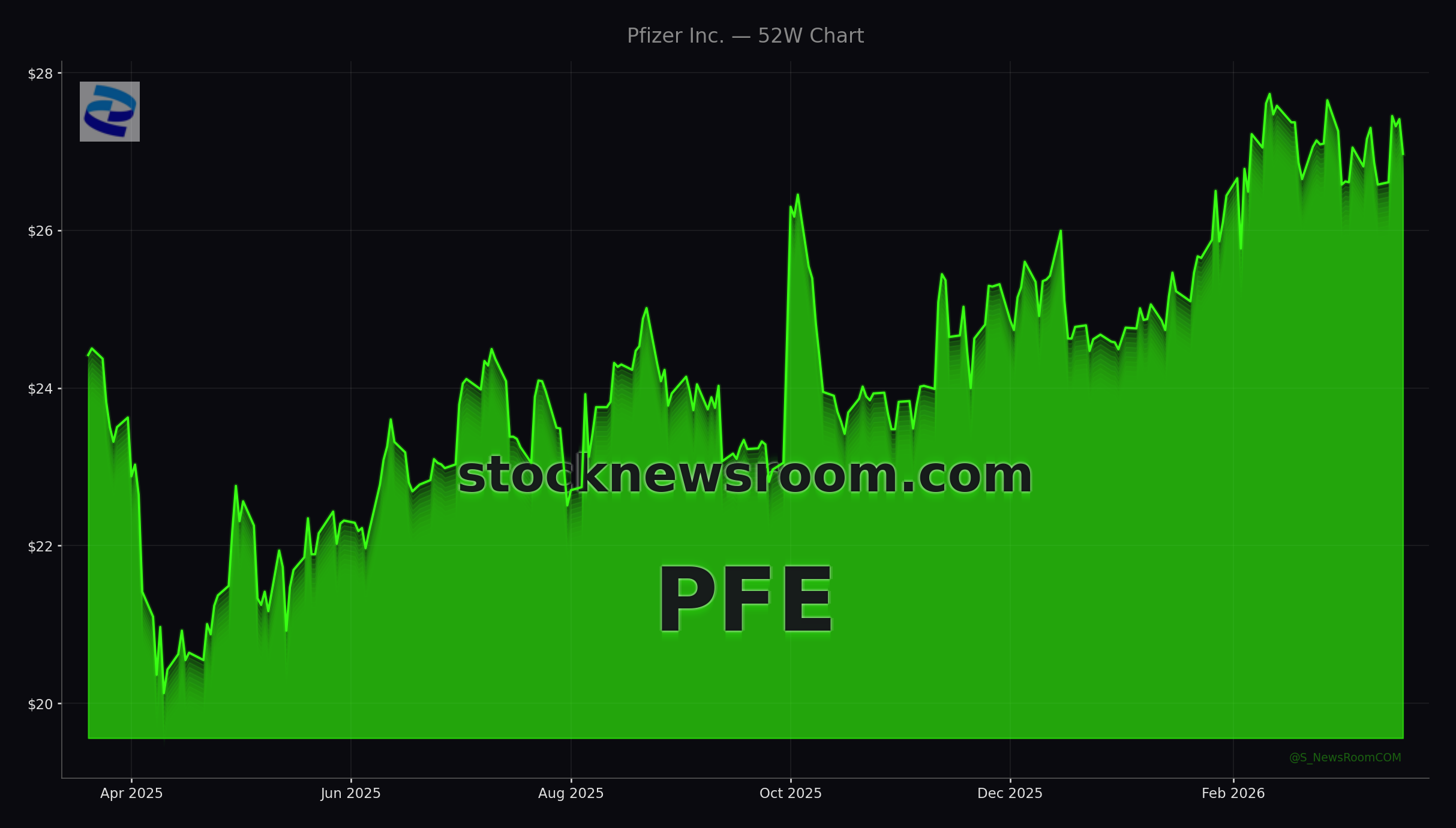
Pfizer shares traded near $26.97 on Monday, slightly below the prior close of $27.03, with pre-market indications around $27.07. The stock remains well under its 52-week high and is broadly viewed as a value and income play after the post-COVID reset in earnings expectations. Several large investors have been repositioning: some institutions cut positions in recent quarters, while others increased exposure on the back of better-than-feared Q4 2025 earnings and pipeline updates in oncology and vaccines.
On Wall Street, consensus remains cautious but not outright bearish. Park Avenue Securities, for example, recently disclosed a larger PFE stake following Pfizer’s Q4 2025 beat, and various firms including Citigroup and Morgan Stanley have highlighted the company’s late-stage pipeline as a key swing factor for the stock’s re-rating. Across coverage, the average target price tracked by MarketBeat centers around the high-$20s, with many analysts holding a “Hold” stance until more clarity emerges on new product launches and cost-cutting benefits.
For long-term investors, the Pfizer Lyme vaccine adds another tangible catalyst to monitor alongside oncology data readouts and potential business development moves. The commercial ramp, if approved, is unlikely to match COVID-era peaks, but it could provide steady, higher-margin revenue that supports the dividend and gradual earnings growth.
How does Pfizer compare with other healthcare plays?
Within the broader healthcare and large-cap pharma group, Pfizer is trying to reposition itself much like Apple did in shifting from a hardware cycle story to a services ecosystem – moving from pandemic windfalls toward a more balanced mix of durable growth drivers. Other vaccine and biotech names, such as Valneva and mid-cap infectious disease specialists, remain much smaller and more volatile, while diversified giants focus on metabolic and oncology franchises. For investors building thematic exposure, that means Lyme disease is one of several differentiated niches, alongside obesity drugs where companies like Eli Lilly and Tesla-style high-growth innovators in other sectors dominate headlines.
Some long-term investors have also grouped Pfizer with high-yield healthcare stocks highlighted in dividend-focused research, which argue that today’s lower valuations and solid balance sheets offer attractive entry points. Against that backdrop, execution on the Pfizer Lyme vaccine launch – from pricing and reimbursement to physician and patient education – will help determine whether the program becomes merely incremental or a visible contributor to the company’s multi-year recovery narrative.
Related coverage
Investors tracking breakthrough therapies across healthcare may also want to look at obesity and metabolic disease pipelines. For example, Eli Lilly’s next-generation GLP-1 and triple-agonist candidates are drawing significant attention after strong late-stage data. An in-depth look at that trend and its implications for the sector can be found in this analysis of Eli Lilly’s retatrutide triple-agonist boom, which explores how powerful Phase 3 diabetes data could reshape expectations for future revenue growth and competition.
Overall, the Phase 3 VALOR results move the Pfizer Lyme vaccine closer to becoming the first modern human Lyme shot, adding a potentially durable asset to Pfizer’s post-COVID vaccine portfolio. For investors, it strengthens the case that the pipeline can gradually refill the company’s growth engine, with upcoming regulatory decisions and launch execution set to determine just how big a role this new vaccine will play in Pfizer’s long-term story.