Will the Novo Nordisk FDA Compounding crackdown finally shut down GLP‑1 copycats and supercharge the branded obesity drug boom?

How does the FDA proposal hit Novo Nordisk?
The core of the Novo Nordisk FDA Compounding story is simple: the FDA now says it sees “no clinical need” for 503B outsourcing facilities to compound semaglutide and similar GLP‑1 drugs from bulk active ingredients when approved products are available. The proposed exclusion covers semaglutide, the engine behind Novo Nordisk’s weight‑loss drug Wegovy and diabetes medicine Ozempic, along with its older GLP‑1 liraglutide. If finalized after a public comment period running into late June, bulk compounders would largely lose the ability to mass‑produce cheaper alternatives unless these drugs land on the FDA’s shortage list.
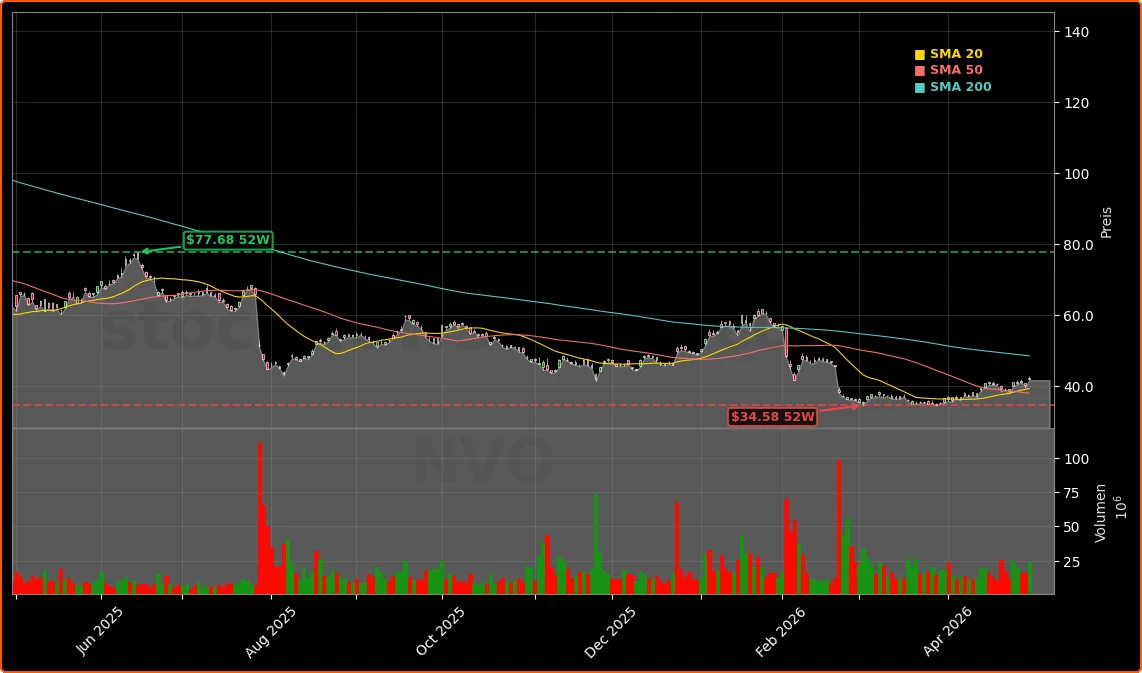
For U.S. and international investors, the timing matters. After a period of supply bottlenecks, Novo Nordisk has poured billions into expanding manufacturing capacity, easing constraints that initially justified some compounding workarounds. The FDA move effectively rewards that investment by narrowing a key gray market channel just as the company’s output is catching up with demand. With NVO now trading well below its 52‑week high but bouncing 4.79% on the day to $42.22, Thursday’s action suggests investors see the proposal as net supportive for pricing power and volume on branded GLP‑1s.
What exactly changes for compounders?
The proposal targets U.S. 503B outsourcing facilities, which can manufacture compounded drugs in bulk with or without individual prescriptions and are primarily overseen by the FDA. These facilities had become a meaningful source of lower‑priced GLP‑1 injections using bulk semaglutide, often marketed as alternatives to Wegovy and Ozempic. Under the new framework, they would be barred from using these bulk active ingredients unless the branded drugs are officially deemed to be in shortage.
Crucially, the measure does not directly affect 503A pharmacies, which prepare customized, patient‑specific compounded products under state regulation. That means a limited compounding channel will remain, but the mass‑production model that threatened to undercut branded pricing could be sharply curtailed. For investors comparing healthcare exposures across sectors, the move is somewhat analogous to how tighter licensing or IP enforcement can benefit leaders like Apple or NVIDIA by limiting lower‑quality clones in their ecosystems.
How does this reshape the GLP‑1 arms race?
Novo Nordisk is not alone in the FDA’s sights. The same proposal would exclude tirzepatide, the active ingredient in Eli Lilly’s weight‑loss shot Zepbound and diabetes drug Mounjaro, from the bulk compounding list. That means the crackdown is broadly market‑wide rather than company‑specific, helping both incumbents defend branded franchises. Still, Novo currently enjoys a time advantage in the oral obesity segment: its Wegovy pill secured a roughly three‑month U.S. head start over Eli Lilly’s rival launch.
Early prescription data for Lilly’s new obesity pill have looked sluggish compared with the Wegovy pill rollout in January, with Leerink Partners analyst David Risinger flagging a more muted start than some on Wall Street expected. Lilly CEO David Ricks has argued that both products are expanding the overall GLP‑1 market, noting that over 80% of new patients were previously untreated with this class. Yet a stricter Novo Nordisk FDA Compounding regime may allow Novo to lean harder on brand recognition and distribution strength while competitors, including Biogen, Merck and Pfizer, explore follow‑on obesity strategies that are still years from full commercialization.
What are analysts watching next?
Novo Nordisk reports earnings next week, and the call is likely to focus heavily on GLP‑1 capacity, pricing, and how management reads the FDA’s evolving stance. While no major Wall Street bank updated its rating on NVO immediately in response to Thursday’s news, investors will be listening for clues that could drive future target changes from firms like Goldman Sachs, Citigroup, Morgan Stanley and RBC Capital Markets. In particular, analysts will want clarity on the scale of previous revenue leakage to compounded semaglutide, and how quickly tighter rules could redirect demand.
From a valuation lens, NVO’s near‑5% move leaves the stock still short of its 52‑week high but reinforces its role as a core healthcare holding for many U.S. and global portfolios, alongside high‑growth names like Tesla or platform champions such as Apple. If management can demonstrate that FDA policy has structurally tilted the playing field back toward approved brands without inviting aggressive political scrutiny on GLP‑1 pricing, multiple expansion could follow.
Related Coverage
Investors looking to place the current Novo Nordisk FDA Compounding debate in a broader strategic context may want to revisit Novo’s push into AI‑enabled drug development and patient support. An in‑depth feature on this theme examines whether a high‑profile collaboration with OpenAI can offset competitive and regulatory pressures in obesity care. For more detail, read Novo Nordisk AI Partnership Boom: Can It Close the Obesity Gap?, which explores how digital tools could reinforce the company’s leadership in GLP‑1 therapies.
When FDA‑approved drugs are available, outsourcing facilities cannot lawfully compound using bulk drug substances unless there is a clear clinical need.— Marty Makary, FDA Commissioner
Altogether, the Novo Nordisk FDA Compounding proposal underscores how regulatory tightening can sometimes favor entrenched innovators. For shareholders, the key questions now are how quickly the rule is finalized and how strongly it boosts branded GLP‑1 demand relative to expectations. The next earnings release and subsequent FDA decisions will show whether this backdrop can sustain NVO’s momentum as a long‑term compounder in health‑care portfolios.