Can the latest Eli Lilly Acquisition in next‑gen cancer therapy really justify its sky‑high valuation beyond the obesity drug boom?

How does the Eli Lilly Acquisition hit the stock?
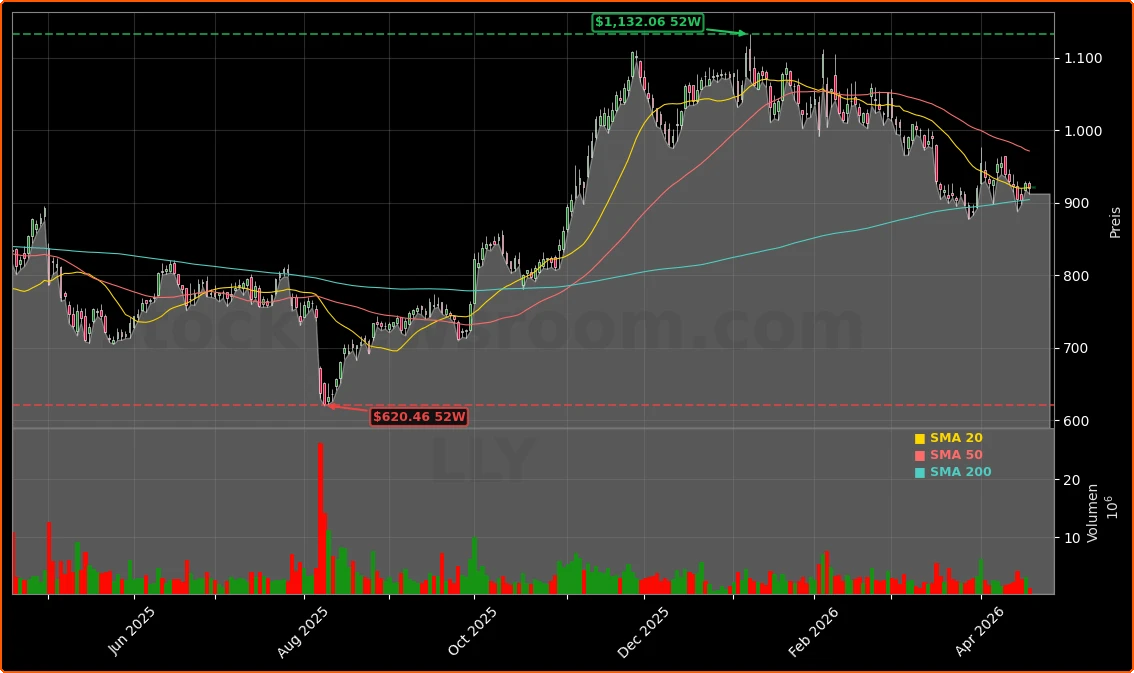
Eli Lilly and Company (LLY) traded at about $919.90 on Monday, slightly below the prior close of $926.50, with after‑hours quotes near $921.50 (all times ET). The stock remains well off its 52‑week high of around $1,133 but far above the 52‑week low near $624, underscoring how much future growth the market has already priced in. Against that backdrop, the latest Eli Lilly Acquisition in oncology and the strong Foundayo data are not just incremental news — they are central to whether the current valuation can be sustained.
For US investors, LLY has become a core healthcare growth name, often compared to mega‑caps like Apple or NVIDIA in terms of index influence. With a market share of roughly 60% in US weight‑loss drugs versus about 39% for Novo Nordisk, the company’s obesity franchise dominates its narrative. Management is now signaling that the next leg of growth will be more balanced, combining metabolic disease leadership with high‑end oncology and gene therapy platforms.
What is Lilly really buying with Kelonia?
The centerpiece of the latest Eli Lilly Acquisition is Kelonia Therapeutics, a private Boston biotech focused on next‑generation CAR‑T gene therapies. Deal terms point to a transaction value north of $2 billion, with some reports in the industry suggesting up to $7 billion including milestones for access to a pipeline targeting blood cancers such as multiple myeloma. Closing is expected in the second half of 2026, subject to regulatory approvals.
Kelonia’s platform is designed to modify a patient’s immune cells directly inside the body, rather than extracting T‑cells and engineering them ex vivo. This in‑vivo CAR‑T approach could dramatically simplify manufacturing, reduce treatment delays, and potentially remove the need for intensive chemotherapy preconditioning. One of Kelonia’s lead candidates is already in early clinical testing in multiple myeloma, but Oppenheimer analysts caution that any approval is likely six to eight years away, underscoring that this Eli Lilly Acquisition is a long‑duration bet rather than an immediate earnings driver.
Lilly is no stranger to big oncology deals. In 2019 it paid about $8 billion for Loxo Oncology, and more recently it agreed to acquire Orna Therapeutics for up to $2.4 billion to bolster its RNA and gene medicine capabilities. The Kelonia transaction fits that pattern: using obesity‑drug cash flows to secure optionality in cancer, inflammatory and genetic diseases for the 2030s and beyond.
How does this reshape Eli Lilly’s cancer strategy?
Oncology has become the second major pillar of Eli Lilly and Company after metabolic disease. Existing drugs such as Verzenio in breast cancer generate substantial revenue, but CAR‑T therapies offer a path into higher‑value, personalized medicine segments currently led by rivals like Johnson & Johnson and Tesla‑like high‑growth innovators in biotech. Management argues that Kelonia’s in‑vivo technology could remove some of the biggest bottlenecks that have limited CAR‑T’s commercial impact: complex logistics, high manufacturing costs, and constrained patient access.
If successful, the technology could be deployed beyond blood cancers into solid tumors, a space where current CAR‑T products have struggled. For US portfolios, that raises the prospect that LLY could eventually command multiple oncology revenue streams, not just one or two flagship drugs. However, with commercialization still years away, near‑term financial contribution will be negligible, and investors should expect continued R&D spending pressure.
Analysts at Morgan Stanley and Goldman Sachs have generally framed Lilly’s recent acquisition spree as strategically sound, emphasizing the need to diversify beyond obesity drugs before pricing or reimbursement headwinds surface. While formal rating changes tied specifically to the Kelonia deal have not yet been flagged, the consensus on Wall Street remains that LLY deserves a growth‑premium multiple relative to more mature pharma peers.
What did the Foundayo cardio study actually show?
Parallel to the Kelonia news, Lilly released pivotal Phase 3 data from the ACHIEVE‑4 trial of its oral GLP‑1 pill Foundayo in more than 2,700 adults with type‑2 diabetes and elevated cardiovascular risk. Compared with insulin glargine, Foundayo cut the risk of major adverse cardiovascular events by about 16%, while an interim analysis showed a roughly 57% reduction in all‑cause mortality. The drug also produced superior weight loss at 52 weeks, with benefits maintained over longer follow‑up, and side effects mainly limited to class‑typical nausea and gastrointestinal issues.
These results directly address prior FDA concerns around potential cardiovascular risk for oral GLP‑1 therapies. Lilly now plans to file for US approval of Foundayo in type‑2 diabetes by the end of Q2 2026. BMO Capital Markets describes the remaining FDA safety requirements as “manageable,” suggesting a relatively clear regulatory path, though timelines for label language and post‑marketing commitments remain key watch points for investors.
Success with Foundayo would deepen Lilly’s GLP‑1 moat against Novo Nordisk and could extend its dominance in oral obesity and diabetes therapies, especially if combination regimens and once‑daily dosing prove attractive to primary‑care physicians. Meanwhile, seven additional Phase 3 trials for the triple‑agonist candidate Retatrutide are expected to read out this year, adding further optionality to the obesity franchise.
Eli Lilly Acquisition strategy vs. Big Pharma peers
Compared with diversified peers like Johnson & Johnson and Pfizer, Lilly is pursuing a more concentrated but higher‑growth portfolio built around obesity, diabetes, oncology and advanced gene therapies. Rather than large, low‑growth consumer health units, Lilly is reinvesting windfall cash into platforms with long‑term upside, much like NVIDIA plowing chip profits back into AI infrastructure. This carries higher execution risk but offers greater potential for earnings acceleration if even a handful of programs succeed.
Citigroup and RBC Capital Markets have both highlighted LLY as a core defensive‑growth holding within the S&P 500 healthcare cohort, citing its obesity pipeline and disciplined capital deployment. The current Eli Lilly Acquisition wave — from Orna to Kelonia and other smaller deals in GI, ophthalmology and gene editing — reinforces that narrative of building a broad innovation stack rather than relying on a single blockbuster.
The next major catalyst is April 30, 2026, when Lilly reports Q1 results and is expected to give more color on Kelonia’s integration, Foundayo’s regulatory timeline and capital allocation priorities. With shares already up solidly over the past 12 months, investor expectations are high, and any hint of delay or safety concern could pressure the stock in the short term.
Related Coverage
For a deeper dive into whether Foundayo can sustain Lilly’s weight‑loss leadership and support today’s valuation, see our detailed analysis of Eli Lilly’s GLP‑1 pill Foundayo and the durability of the current obesity drug boom. The article examines competitive dynamics with Novo Nordisk and what different pricing scenarios could mean for long‑term margins.
Investors comparing healthcare exposure across large caps may also want to read our coverage of Johnson & Johnson’s latest earnings beat and dividend hike, which discusses how a more diversified pharma and medical devices portfolio stacks up against Lilly’s higher‑growth, higher‑risk strategy.
Kelonia’s in‑vivo CAR‑T platform is a long‑duration bet that could eventually transform access to cell therapies and reshape Eli Lilly’s oncology mix.— Healthcare strategist at a New York asset manager
Overall, the latest Eli Lilly Acquisition in CAR‑T and the positive Foundayo data underline Lilly’s ambition to pair near‑term obesity cash flows with long‑dated oncology innovation. For US investors, that combination keeps LLY firmly positioned as a premium growth name within healthcare. The coming quarters — starting with the April 30 earnings update — will show whether execution can keep pace with expectations embedded in the share price.