Is the latest FDA shift on peptides turning Hims & Hers into a long-term growth story or just fueling a speculative spike?

Why is Hims & Hers peptide regulation moving the stock?
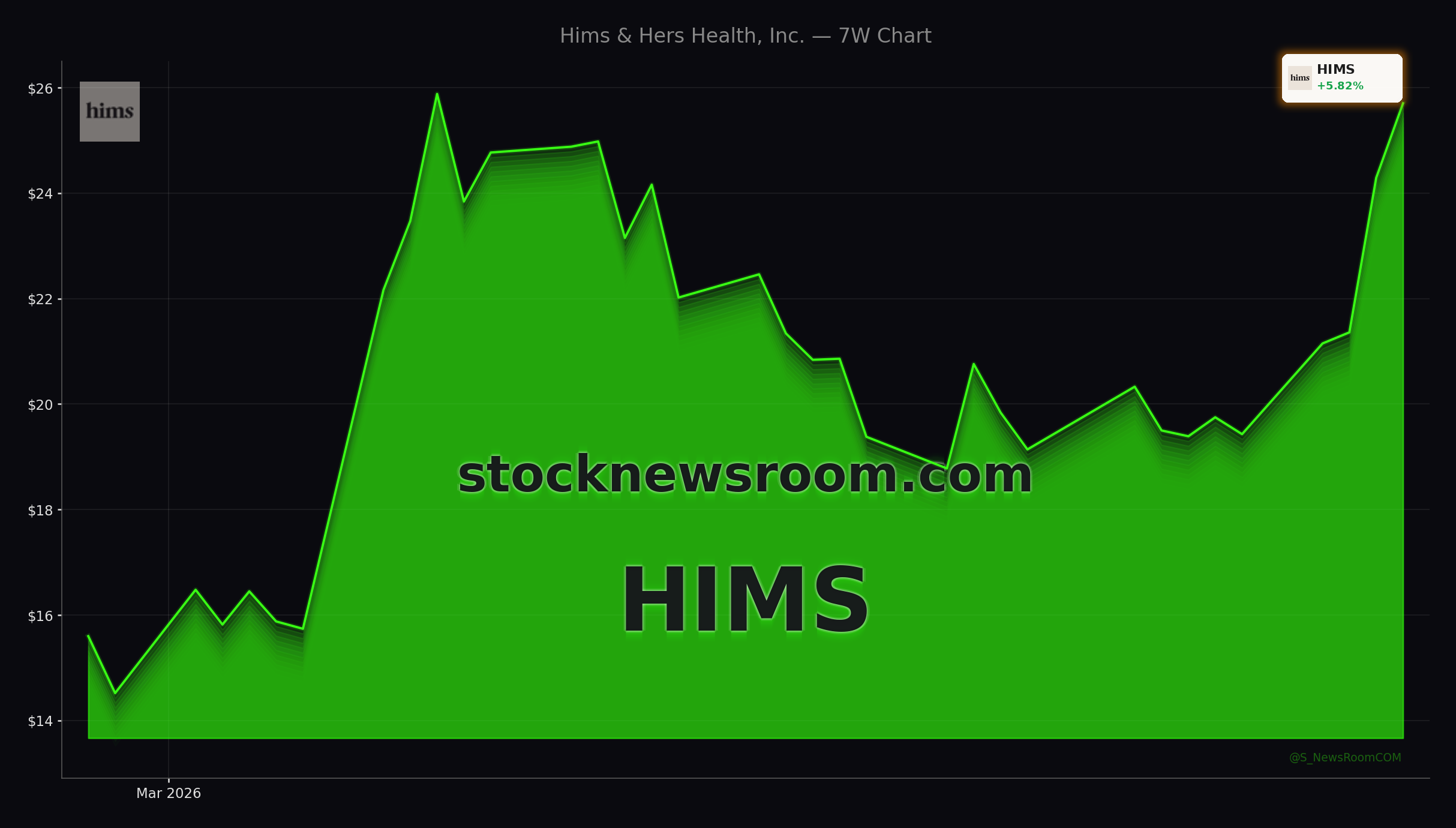
Hims & Hers Health is rallying after U.S. Health Secretary RFK Jr. indicated that the FDA is working to remove or soften so‑called Category 2 restrictions on roughly a dozen peptides. These compounds, which have gained popularity for weight loss, muscle regeneration and broader wellness, have been constrained by guidance that has limited how they can be prescribed and compounded. Any move to clarify or ease those rules would directly benefit platforms that can scale compliant manufacturing and digital distribution.
The timing is favorable for Hims & Hers. The company recently acquired and ramped a specialized manufacturing facility in California focused on peptide production, complementing its existing GLP‑1 infrastructure. With HIMS shares now trading more than 10% higher on the session and well above their 20‑day moving average, traders are treating Hims & Hers peptide regulation as a concrete catalyst, not just a policy headline. The market is effectively assigning value to optionality: if these peptides can be legally and reliably offered through telehealth, Hims & Hers is positioned to be an early mover.
That optionality matters in a U.S. market where wellness, longevity and medical weight loss trends are pulling billions in consumer spending toward platforms that combine digital access with pharmacy capabilities. While large‑cap healthcare names like Johnson & Johnson and Apple-adjacent health initiatives in wearables capture headlines, smaller, fast‑growing players such as Hims & Hers can respond more quickly to incremental regulatory openings.
How is Wall Street reacting to Hims & Hers?
Analysts are updating their models in response to the evolving regulatory backdrop. Bank of America Securities, led by analyst Alan Lutz, recently raised its price target on Hims & Hers from $21 to $25 while maintaining a Neutral rating. Lutz frames the FDA’s peptide move as an early but positive step, highlighting that it could allow Hims & Hers to repurpose some GLP‑1 compounding capacity toward new peptide therapies focused on longevity, sleep and preventative medicine. The raised target reflects this added strategic flexibility rather than a full‑blown re‑rating.
Other firms are more constructive. Barclays keeps an Overweight rating on Hims & Hers with a $29 price target, pointing to rapid subscriber growth and strong revenue momentum. Leer Inc. rates the stock Market Perform with a $25 target, roughly in line with Bank of America’s new level, signaling a more balanced risk‑reward view at current prices. Together, these calls underscore a divide on Wall Street: some see Hims & Hers peptide regulation as a potential multi‑year growth driver, while others caution that near‑term valuation and margin pressure still matter.
Operationally, Hims & Hers ended 2025 with more than 2.5 million subscribers, up 13% year over year, and average revenue per subscriber of $83 per month, up 11%. Full‑year 2025 revenue reached about $2.35 billion, a 59% jump, with management guiding to $2.7–$2.9 billion in 2026. Those are growth rates that stand out even against tech‑enabled healthcare peers on the NASDAQ and S&P 500, including digital health platforms backed by giants like NVIDIA in AI infrastructure and Tesla-style direct‑to‑consumer branding strategies.
Does the peptide pivot solve margin and cash flow risks?
Despite the enthusiasm around Hims & Hers peptide regulation, there are real financial trade‑offs. The company’s thesis for 2026 still leans heavily on continued access to compounded semaglutide and other GLP‑1‑related treatments. That exposure has already pressured gross margins, which have compressed by roughly 500 basis points year over year as the business scaled branded weight‑loss therapies. Free cash flow dipped slightly negative in Q4 2025 at around -$2.57 million, a signal that aggressive reinvestment and product mix shifts can strain near‑term profitability.
If FDA guidance ultimately enables a portfolio of wellness peptides beyond GLP‑1, Hims & Hers could diversify revenue and improve its operating leverage. The California facility gives it in‑house manufacturing control, which may support better margins over time versus relying solely on third‑party supply. Management has also outlined ambitions for a longevity specialty offering, bundling peptides, coenzymes and GLP/GIP treatments through its telehealth platform.
For U.S. investors, the question is whether this regulatory inflection will be large enough and fast enough to offset margin pressure and any future tightening in GLP‑1 oversight. Compared with diversified healthcare heavyweights like Johnson & Johnson or tech‑driven consumer ecosystems led by Apple, Hims & Hers remains a focused, higher‑beta name whose trajectory is unusually sensitive to FDA interpretations and reimbursement dynamics.
Related Coverage
Investors tracking Hims & Hers peptide regulation should also look at how the company is building its weight‑loss franchise more broadly. A recent deep dive explores whether its GLP‑1 collaboration with Novo Nordisk can turn telehealth‑based obesity care into the next growth engine; details are available in this analysis of the Hims & Hers GLP-1 partnership and the emerging weight-loss boom. For a wider healthcare context, blue-chip earnings can provide a useful benchmark: this review of Johnson & Johnson’s latest earnings surge and dividend hike breaks down how a mature S&P 500 constituent balances growth, cash returns and valuation—helping investors frame the risk‑reward of faster‑growing names like Hims & Hers.
In summary, Hims & Hers peptide regulation is emerging as a genuine catalyst, lifting the stock as Washington signals a willingness to revisit peptide rules and Bank of America and others adjust their targets. For U.S. investors, the story is less about today’s 10% move and more about whether new peptide therapies can broaden the company’s revenue base and eventually support margin expansion. The next stages of FDA review and management’s execution on its peptide manufacturing footprint will determine whether this regulatory tailwind turns into a durable growth driver for long‑term portfolios.